Difference between revisions of "Part:BBa K2621009"
| (One intermediate revision by the same user not shown) | |||
| Line 3: | Line 3: | ||
<partinfo>BBa_K2621009 short</partinfo> | <partinfo>BBa_K2621009 short</partinfo> | ||
| − | + | [[Image:T--Vilnius-Lithuania-OG--catseqsmalllogo.jpeg|right|200px|<b>Figure 1. </b> Abstract scheme of the Catalytic Activity Sequencing]] | |
| + | |||
| + | This part is a computationally derived mutant of the [[https://parts.igem.org/Part:BBa_K2621000 CAT-Seq Esterase]]. You can read more about how the mutants were derived [[http://2018.igem.org/Team:Vilnius-Lithuania-OG/Model by following this link]]. The goal of the mutant development was to obtain Esterase variants with different activities. The reason for that was to create a small different activity library of the CAT-Seq esterase in order to assess the accuracy of CAT-Seq when comparing its results to standard and well established low throughput methods. | ||
| + | |||
| + | CAT-Seq Esterase is a hydrolase that was used in Catalytic Activity Sequencing system for its promiscuous capability to hydrolyse N4-acyl-2'-deoxycytidine triphosphates (<b>Substrate Nucleotides</b>). It has been found that the enzyme accepts various N4-acyl-2'-deoxycytidine triphosphates as substrates for hydrolysis, leaving a 2'-deoxycytidine triphosphate (<b>Product Nucleotide</b>). | ||
| + | |||
| + | Sequence-wise it is the most similar to <b>tannases</b> (EC 3.1.1.20) and <b>feruloyl esterases</b> (EC 3.1.1.73), however to date there has been no enzyme characterized hydrolysing N4-amidic bond in modified deoxycytidine triphosphates. | ||
| + | |||
| + | It is the main component of Catalytic Activity Sequencing (CAT-Seq) method. CAT-Seq is a method for high-throughput catalytic biomolecule and genetic regulatory part <b>activity-sequence relationship</b> assessment toolkit. | ||
| + | |||
| + | |||
| + | |||
| + | See how this part is used in the CAT-Seq [[#About CAT-Seq|by pressing here!]] | ||
| − | |||
| − | |||
| − | |||
<span class='h3bb'>Sequence and Features</span> | <span class='h3bb'>Sequence and Features</span> | ||
<partinfo>BBa_K2621009 SequenceAndFeatures</partinfo> | <partinfo>BBa_K2621009 SequenceAndFeatures</partinfo> | ||
| Line 17: | Line 26: | ||
<partinfo>BBa_K2621009 parameters</partinfo> | <partinfo>BBa_K2621009 parameters</partinfo> | ||
<!-- --> | <!-- --> | ||
| + | |||
| + | __TOC__ | ||
| + | |||
| + | |||
| + | |||
| + | =Introduction= | ||
| + | ==Biology== | ||
| + | ===Description of the CAT-Seq esterase=== | ||
| + | |||
| + | |||
| + | <b>CAT-Seq Esterase</b> is a hydrolase that has a promiscuous capability to hydrolyse N4-acyl-2'-deoxycytidine triphosphates (<b>Substrate Nucleotide</b>). Sequence-wise it is the most similar to tannases (EC 3.1.1.20) and feruloyl esterases (EC 3.1.1.73), however to date there has been no enzyme characterized hydrolysing N4-amidic bond in modified deoxycytidine triphosphates. | ||
| + | |||
| + | To further elaborate on the fact, it has been found that the enzyme accepts various N4-acyl-2'-deoxycytidine triphosphates as substrates for hydrolysis, leaving a 2'-deoxycytidine triphosphate (<b>Product Nucleotide</b>). | ||
| + | |||
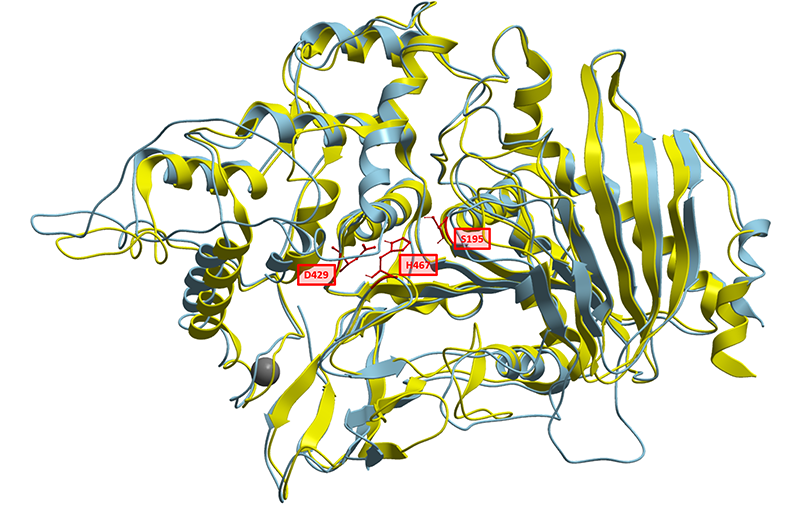
| + | To better understand the nature of this biological part, we have generated a <b>structural homology model</b> based on solved structures of homologous feruloyl esterases. Currently known feruloyl esterases contain a <b>catalytic triad</b> in their active sites consisting of Serine, Aspartate and Histidine. Aligning CAT-Seq Esterase to the structural 3WMT template, we could also identify the catalytic triad consisting of <b>Ser195</b>, <b>Asp429</b>, <b>His467</b>. The sequence similarities and the positions Catalytic Triad amino acids are highlighted in the illustration below (Fig. 1). | ||
| + | |||
| + | [[Image:T--Vilnius-Lithuania-OG--structuremodel1.png|center|frame|300px|<b>Figure 1. </b> Superposition of the generated homology model (<b>light blue</b>) and the template used (<b>yellow</b>, PDB ID: 6G21). Catalytic triad residues are marked in red boxes with numbering. Calcium ion shown as grey sphere.]] | ||
| + | |||
| + | ===In-Silico design of the CAT-Seq esterase mutants=== | ||
| + | |||
| + | The reason of making esterase mutants was <b>to benchmark the accuracy</b> of CAT-Seq system. We have aimed to make Esterase mutants that would have slightly <b>different</b> activities. After deriving the mutations, we have measured their activities using <b>standard methods</b>, and then compared them with CAT-Seq measurements. Therefore, the esterase and its mutants can now be used for <b>accuracy calibration</b> when setting up the CAT-Seq for the first time. | ||
| + | |||
| + | The residues chosen for mutation were scattered around the <b>catalytic triad</b> of the esterase and involved mainly <b>polar and aromatic</b> side chains. In literature, some of these side chains are assumed to form bonds stabilizing the binding of the substrate (Fig. 2). | ||
| + | |||
| + | |||
| + | [[Image:T--Vilnius-Lithuania-OG--mutationscheme2.jpeg|center|frame|300px|<b>Figure 2. <b>View of the CAT-Seq Esterase Active Center</b>. Residue coloring: <b>catalytic triad</b> - red, <b>mutation positions</b> - orange, <b>calcium-coordinating residues</b> - cyan. Catalytic residues as well as successful mutations are numbered in boxes. Hydrogen bonds in the catalytic triad represented as black dashed lines.]] | ||
| + | |||
| + | |||
| + | After carefully selecting the mutations in-silico, we have synthesized and measured the Activity of those esterase mutants in the laboratory. The <b>mutations have successfully altered the activity</b> of the original Esterase. Also, there were no cases of complete inactivation of enzyme catalytic activity from the mutants we managed test. Please head to the results section for further information. | ||
| + | |||
| + | ==Usage with CAT-Seq (Catalytic Activity Sequencing)== | ||
| + | |||
| + | ===About CAT-Seq=== | ||
| + | [[Image:T--Vilnius-Lithuania-OG--registrycatseq.jpeg|center|T--Vilnius-Lithuania-OG--registrycatseq.jpeg|350px|<b>Figure 1. </b> Abstract scheme of the Catalytic Activity Sequencing method]] | ||
| + | |||
| + | CAT-Seq stands for <b>Catalytic Activity Sequencing</b> - a system designed and built for high-speed activity and interaction characterization of Catalytic and Regulatory biological parts. You can learn more about CAT-Seq [http://2018.igem.org/Team:Vilnius-Lithuania-OG by clicking this link] | ||
| + | |||
| + | ===Catalytic Activity Sequencing Overview=== | ||
| + | [[Image:T--Vilnius-Lithuania-OG--catseqbtestasviensdutrysiomolecule.jpeg|center|frame|300px|<b>Figure 1. </b> Abstract scheme of the Catalytic Activity Sequencing method]] | ||
| + | |||
| + | #<b>Library preparation</b> - A library of catalytic biomolecules is prepared. | ||
| + | #<b>Library encapsulation into droplets</b> - Every library fragment is physically separated by encapsulating them into picoliter water droplets. Also, substrate nucleotides, the targets for catalytic biomolecules, are encapsulated. | ||
| + | #<b>Catalytic biomolecule production</b> - In each droplet catalytic biomolecules are produced. | ||
| + | #<b>Catalysis of the substrate conversion</b> - Catalytic biomolecules may recognise the Substrate Nucleotides as a target for chemical reaction catalysis. Depending on biomolecule activity, a specific number of nucleotides with removed substrates (product nucleotides) is established in each droplet. | ||
| + | #<b>Activity Recording</b> | ||
| + | ##<b>Droplet Merging</b> - each of prior droplet is merged with new droplet that contains DNA amplification mix and reference nucleotides. The reference nucleotides are helping to tracking the Product Nucleotide number. | ||
| + | ##<b>DNA amplification</b> - DNA is amplified using the different unique catalytic biomolecule DNA in each droplet. During the amplification, the Product Nucleotides and the Reference Nucleotides are incorporated into the DNA sequence. | ||
| + | #<b>Activity Reading by Nanopore Sequencing</b> - All of the droplets are broken and the amplified DNA is sequenced. During the sequencing, biomolecule’s activity is retrieved by calculating reference and Product Nucleotides (substrate removed), together with the sequence of particular biomolecule variant. | ||
| + | |||
| + | ===Determining the accuracy of CAT-Seq=== | ||
| + | Trying to build a CAT-Seq pipeline in your own laboratory will require the CAT-Seq esterase in order to troubleshoot the system and assess the measurement accuracy and precision. In other words, the esterase and its mutants can be used to calibrate the CAT-Seq. | ||
| + | |||
| + | Together with the Esterase, its substrate attached to a nucleotide is required (substrate nucleotide). In the standard case, the <b>Substrate Nucleotide</b> is N4-benzoyl-2'-deoxycytidine triphosphate. If the Esterase catalyzes the removal of the substrate from the nucleotide, it becomes the <b>Product Nucleotide - </b> 2'-deoxycytidine triphosphate. | ||
| + | |||
| + | =Part Characterization (Vilnius-Lithuania Overgraduate 2018)= | ||
| + | ==Kinetic characterization of the original CAT-Seq Esterase == | ||
| + | |||
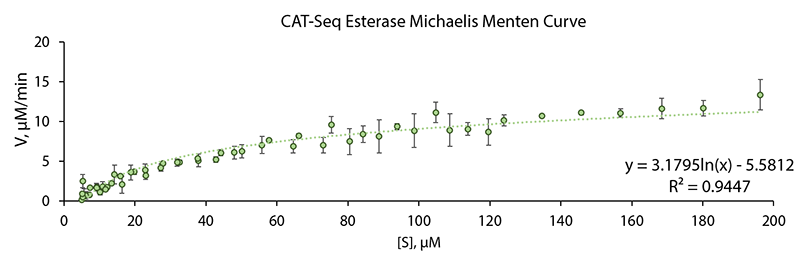
| + | Spectrofotometic kinetic data based on decay of absorbance at 310 nm due to substrate nucleotide catalytic conversion was gathered using a range of starting substrate nucleotide concentrations. A Michelis Menten curve for the CAT-Seq esterase was plotted using the aquired data. Data shown in 1 Fig., show perfect fit (R2 = 0.9449) to a standard Michaelis Menten curve. | ||
| + | |||
| + | [[Image:T--Vilnius-Lithuania-OG--menten1.jpeg|center|frame|250px|<b>Figure 1. Michaelis Menten Curve generated for CAT-Seq Esterase enzyme.</b> The initial velocity of the enzyme was determined spectrophotometrically as a decay of absorbance at 310 nm at different starting substrate nucleotide concentrations. The graph shows the average values of three independent experiments.]] | ||
| + | |||
| + | Michaelis Menten plot transformations were generated for CAT-Seq esterase enzyme ([https://parts.igem.org/Part:BBa_K2621000 BBa_K2621000]). 2 Fig., display Lineweaver-Burk and Hanes-Woolf transformation plot. Based on the equation koeficients in the Hanes-Woolf transformation, the expermentally determined Vmax value is <b>17.2 µM/min</b>, Km value is <b>86 µM</b>. | ||
| + | |||
| + | |||
| + | [[Image:T--Vilnius-Lithuania-OG--menten2.jpeg|center|frame|250px|<b>Figure 1. Two Michaelis Menten Curve transformations for CAT-Seq Esterase enzyme.</b> Lineweaver-Burk and Hanes-Woolf transformations for Michaelis Menten curve generated earlier. Both of the transformations were performed on data acquired from 3 independent experiments. Graphs show the fitted linear function and its correlation coefficient.]] | ||
| + | |||
| + | ==Esterase and its mutants catalytic activity determination== | ||
| + | |||
| + | The 10 esterase mutants housing mutations at bioinformatically predicted sites were created. To see why and how mutations were <b>designed in-silico</b>, [http://2018.igem.org/Team:Vilnius-Lithuania-OG/Model please click this link]. Each of the mutant was constructed utilizing PCR and synthesized using In vitro transcription and translation kit and their catalytic activity towards N4-benzoyl-2'-deoxycytidine triphosphate were tested. | ||
| + | |||
| + | [[Image:T--Vilnius-Lithuania-OG--mutant3.jpeg|center|frame|300px|<b>Figure 3. Relative activity of In silico generated CAT-Seq esterase mutants</b> CAT-Seq esterase mutant sequences were generated In silico and synthesised using in vitro transcription and translation kit. The graph shows the relative catalytic activity of each generated mutant measured spectrophotometrically and the corresponding mutation site.The activity was normalized to wild type (WT) enzyme. ]] | ||
| + | |||
| + | |||
| + | The reaction kinetics were measured using the spectrophotometer as a decrease of absorbance. Figure 3 displays the relative hydrolysis speed of each mutant generated. As seen from these results, a variety of mutants, showing different catalysis speeds were produced. Some of the amino acids changes affected the activity drastically, for example Trp224 to Tyr, Lys227 to Arg or Glu509 to Lys. Other mutations only modulated the activity Asn107 to Asp or Glu194 to Ala. Additionally, large 8 amino acid deletion at position Pro348-Hy356 caused only a moderate decrease in enzymes activity. | ||
| + | |||
| + | ==Esterase and mutants activity assessment using CAT-Seq== | ||
| + | |||
| + | The constructed in silico designed mutant library was subjected to catalytic activity sequencing. By applying the data preparation and analysis pipeline, the mean methylation scores arising from different ratios of catalytically converted and reference nucleotides for each barcoded mutant DNA template were filtered and extracted from the DNA embedded with catalytic activity information (in a form of incorporated reference to catalytically converted nucleotide ratio). The collected data was normalized over Wild Type Esterase and K227R mutant (lowest activity). | ||
| + | |||
| + | [[Image:T--Vilnius-Lithuania-OG--catorbswefwefmolecule.jpeg|center|frame|300px|<b>Figure 1. Comparison of In bulk and CAT-Seq measured esterase mutant relative activity. </b> In silico generated Esterase mutant library was subjected to catalytic activity sequencing. The mean methylation scores for each barcoded mutant DNA template were filtered and extracted. The collected data was normalized over Wild Type CAT seq Esterase and K227R mutant (lowest activity). The relative activity, extracted from the mean methylation score of each mutant read is compared to measurement data gathered in standard sized reactions (in bulk) .]] | ||
| + | |||
| + | |||
| + | The relative methylation score (reference nucleotide count) of each mutant read corresponds to the activity of the enzyme it encodes. The higher the activity of the expressed enzyme, the lower methylation score are assigned, due to catalytic conversion of substrate nucleotides. The comparison of the results, gathered with CAT-Seq catalytic activity sequencing method and in standard sized reactions (in bulk) spectrophotometric data (Fig 1.) conclude the viability of CAT-Seq approach. The activity reading, extracted from the DNA sequence correlates with the in bulk measurement data perfectly. The activity of the each Esterase mutant is measured accurately and is assigned to the corresponding DNA sequence. | ||
Latest revision as of 19:03, 17 October 2018
CAT-Seq Esterase Mutant (W224Y)
This part is a computationally derived mutant of the [CAT-Seq Esterase]. You can read more about how the mutants were derived http://2018.igem.org/Team:Vilnius-Lithuania-OG/Model by following this link. The goal of the mutant development was to obtain Esterase variants with different activities. The reason for that was to create a small different activity library of the CAT-Seq esterase in order to assess the accuracy of CAT-Seq when comparing its results to standard and well established low throughput methods.
CAT-Seq Esterase is a hydrolase that was used in Catalytic Activity Sequencing system for its promiscuous capability to hydrolyse N4-acyl-2'-deoxycytidine triphosphates (Substrate Nucleotides). It has been found that the enzyme accepts various N4-acyl-2'-deoxycytidine triphosphates as substrates for hydrolysis, leaving a 2'-deoxycytidine triphosphate (Product Nucleotide).
Sequence-wise it is the most similar to tannases (EC 3.1.1.20) and feruloyl esterases (EC 3.1.1.73), however to date there has been no enzyme characterized hydrolysing N4-amidic bond in modified deoxycytidine triphosphates.
It is the main component of Catalytic Activity Sequencing (CAT-Seq) method. CAT-Seq is a method for high-throughput catalytic biomolecule and genetic regulatory part activity-sequence relationship assessment toolkit.
See how this part is used in the CAT-Seq by pressing here!
Sequence and Features
- 10COMPATIBLE WITH RFC[10]
- 12COMPATIBLE WITH RFC[12]
- 21COMPATIBLE WITH RFC[21]
- 23COMPATIBLE WITH RFC[23]
- 25INCOMPATIBLE WITH RFC[25]Illegal NgoMIV site found at 487
- 1000COMPATIBLE WITH RFC[1000]
Introduction
Biology
Description of the CAT-Seq esterase
CAT-Seq Esterase is a hydrolase that has a promiscuous capability to hydrolyse N4-acyl-2'-deoxycytidine triphosphates (Substrate Nucleotide). Sequence-wise it is the most similar to tannases (EC 3.1.1.20) and feruloyl esterases (EC 3.1.1.73), however to date there has been no enzyme characterized hydrolysing N4-amidic bond in modified deoxycytidine triphosphates.
To further elaborate on the fact, it has been found that the enzyme accepts various N4-acyl-2'-deoxycytidine triphosphates as substrates for hydrolysis, leaving a 2'-deoxycytidine triphosphate (Product Nucleotide).
To better understand the nature of this biological part, we have generated a structural homology model based on solved structures of homologous feruloyl esterases. Currently known feruloyl esterases contain a catalytic triad in their active sites consisting of Serine, Aspartate and Histidine. Aligning CAT-Seq Esterase to the structural 3WMT template, we could also identify the catalytic triad consisting of Ser195, Asp429, His467. The sequence similarities and the positions Catalytic Triad amino acids are highlighted in the illustration below (Fig. 1).
In-Silico design of the CAT-Seq esterase mutants
The reason of making esterase mutants was to benchmark the accuracy of CAT-Seq system. We have aimed to make Esterase mutants that would have slightly different activities. After deriving the mutations, we have measured their activities using standard methods, and then compared them with CAT-Seq measurements. Therefore, the esterase and its mutants can now be used for accuracy calibration when setting up the CAT-Seq for the first time.
The residues chosen for mutation were scattered around the catalytic triad of the esterase and involved mainly polar and aromatic side chains. In literature, some of these side chains are assumed to form bonds stabilizing the binding of the substrate (Fig. 2).

After carefully selecting the mutations in-silico, we have synthesized and measured the Activity of those esterase mutants in the laboratory. The mutations have successfully altered the activity of the original Esterase. Also, there were no cases of complete inactivation of enzyme catalytic activity from the mutants we managed test. Please head to the results section for further information.
Usage with CAT-Seq (Catalytic Activity Sequencing)
About CAT-Seq
CAT-Seq stands for Catalytic Activity Sequencing - a system designed and built for high-speed activity and interaction characterization of Catalytic and Regulatory biological parts. You can learn more about CAT-Seq [http://2018.igem.org/Team:Vilnius-Lithuania-OG by clicking this link]
Catalytic Activity Sequencing Overview
- Library preparation - A library of catalytic biomolecules is prepared.
- Library encapsulation into droplets - Every library fragment is physically separated by encapsulating them into picoliter water droplets. Also, substrate nucleotides, the targets for catalytic biomolecules, are encapsulated.
- Catalytic biomolecule production - In each droplet catalytic biomolecules are produced.
- Catalysis of the substrate conversion - Catalytic biomolecules may recognise the Substrate Nucleotides as a target for chemical reaction catalysis. Depending on biomolecule activity, a specific number of nucleotides with removed substrates (product nucleotides) is established in each droplet.
- Activity Recording
- Droplet Merging - each of prior droplet is merged with new droplet that contains DNA amplification mix and reference nucleotides. The reference nucleotides are helping to tracking the Product Nucleotide number.
- DNA amplification - DNA is amplified using the different unique catalytic biomolecule DNA in each droplet. During the amplification, the Product Nucleotides and the Reference Nucleotides are incorporated into the DNA sequence.
- Activity Reading by Nanopore Sequencing - All of the droplets are broken and the amplified DNA is sequenced. During the sequencing, biomolecule’s activity is retrieved by calculating reference and Product Nucleotides (substrate removed), together with the sequence of particular biomolecule variant.
Determining the accuracy of CAT-Seq
Trying to build a CAT-Seq pipeline in your own laboratory will require the CAT-Seq esterase in order to troubleshoot the system and assess the measurement accuracy and precision. In other words, the esterase and its mutants can be used to calibrate the CAT-Seq.
Together with the Esterase, its substrate attached to a nucleotide is required (substrate nucleotide). In the standard case, the Substrate Nucleotide is N4-benzoyl-2'-deoxycytidine triphosphate. If the Esterase catalyzes the removal of the substrate from the nucleotide, it becomes the Product Nucleotide - 2'-deoxycytidine triphosphate.
Part Characterization (Vilnius-Lithuania Overgraduate 2018)
Kinetic characterization of the original CAT-Seq Esterase
Spectrofotometic kinetic data based on decay of absorbance at 310 nm due to substrate nucleotide catalytic conversion was gathered using a range of starting substrate nucleotide concentrations. A Michelis Menten curve for the CAT-Seq esterase was plotted using the aquired data. Data shown in 1 Fig., show perfect fit (R2 = 0.9449) to a standard Michaelis Menten curve.
Michaelis Menten plot transformations were generated for CAT-Seq esterase enzyme (BBa_K2621000). 2 Fig., display Lineweaver-Burk and Hanes-Woolf transformation plot. Based on the equation koeficients in the Hanes-Woolf transformation, the expermentally determined Vmax value is 17.2 µM/min, Km value is 86 µM.

Esterase and its mutants catalytic activity determination
The 10 esterase mutants housing mutations at bioinformatically predicted sites were created. To see why and how mutations were designed in-silico, [http://2018.igem.org/Team:Vilnius-Lithuania-OG/Model please click this link]. Each of the mutant was constructed utilizing PCR and synthesized using In vitro transcription and translation kit and their catalytic activity towards N4-benzoyl-2'-deoxycytidine triphosphate were tested.

The reaction kinetics were measured using the spectrophotometer as a decrease of absorbance. Figure 3 displays the relative hydrolysis speed of each mutant generated. As seen from these results, a variety of mutants, showing different catalysis speeds were produced. Some of the amino acids changes affected the activity drastically, for example Trp224 to Tyr, Lys227 to Arg or Glu509 to Lys. Other mutations only modulated the activity Asn107 to Asp or Glu194 to Ala. Additionally, large 8 amino acid deletion at position Pro348-Hy356 caused only a moderate decrease in enzymes activity.
Esterase and mutants activity assessment using CAT-Seq
The constructed in silico designed mutant library was subjected to catalytic activity sequencing. By applying the data preparation and analysis pipeline, the mean methylation scores arising from different ratios of catalytically converted and reference nucleotides for each barcoded mutant DNA template were filtered and extracted from the DNA embedded with catalytic activity information (in a form of incorporated reference to catalytically converted nucleotide ratio). The collected data was normalized over Wild Type Esterase and K227R mutant (lowest activity).

The relative methylation score (reference nucleotide count) of each mutant read corresponds to the activity of the enzyme it encodes. The higher the activity of the expressed enzyme, the lower methylation score are assigned, due to catalytic conversion of substrate nucleotides. The comparison of the results, gathered with CAT-Seq catalytic activity sequencing method and in standard sized reactions (in bulk) spectrophotometric data (Fig 1.) conclude the viability of CAT-Seq approach. The activity reading, extracted from the DNA sequence correlates with the in bulk measurement data perfectly. The activity of the each Esterase mutant is measured accurately and is assigned to the corresponding DNA sequence.