Difference between revisions of "Part:BBa K1321359"
| (4 intermediate revisions by 2 users not shown) | |||
| Line 1: | Line 1: | ||
| − | |||
__NOTOC__ | __NOTOC__ | ||
<partinfo>BBa_K1321359 short</partinfo> | <partinfo>BBa_K1321359 short</partinfo> | ||
| − | + | A LacI-promoter expression construct of super-folder GFP fused N-terminally to dCBD (a cellulose-binding domain), which contains an endogenous N-terminal linker sequence. | |
| + | |||
| + | [[File:IC-2014_glow1.jpg |200px|left|]] | ||
| + | |||
| + | |||
| + | This construct is part of a library of Super-folder GFP fusions with cellulose binding domains, which we used to assay the CBD binding affinity. Please see our [http://2014.igem.org/Team:Imperial/Functionalisation project page] for more information. The collection of sfGFP-CBD fusion parts can be seen in the table below: [[File:IC14-sfGFP-part-table.PNG]] | ||
| + | |||
| + | Note that the stop codon plus 6 bp at the end of the sequence are included the RFC25 suffix which is not shown. The prefix to this part is RFC10 format. | ||
| + | |||
| + | As part of our project we carried out an assay to determine the relative binding ability of CBD-sfGFP fusions to bacterial cellulose. | ||
| + | |||
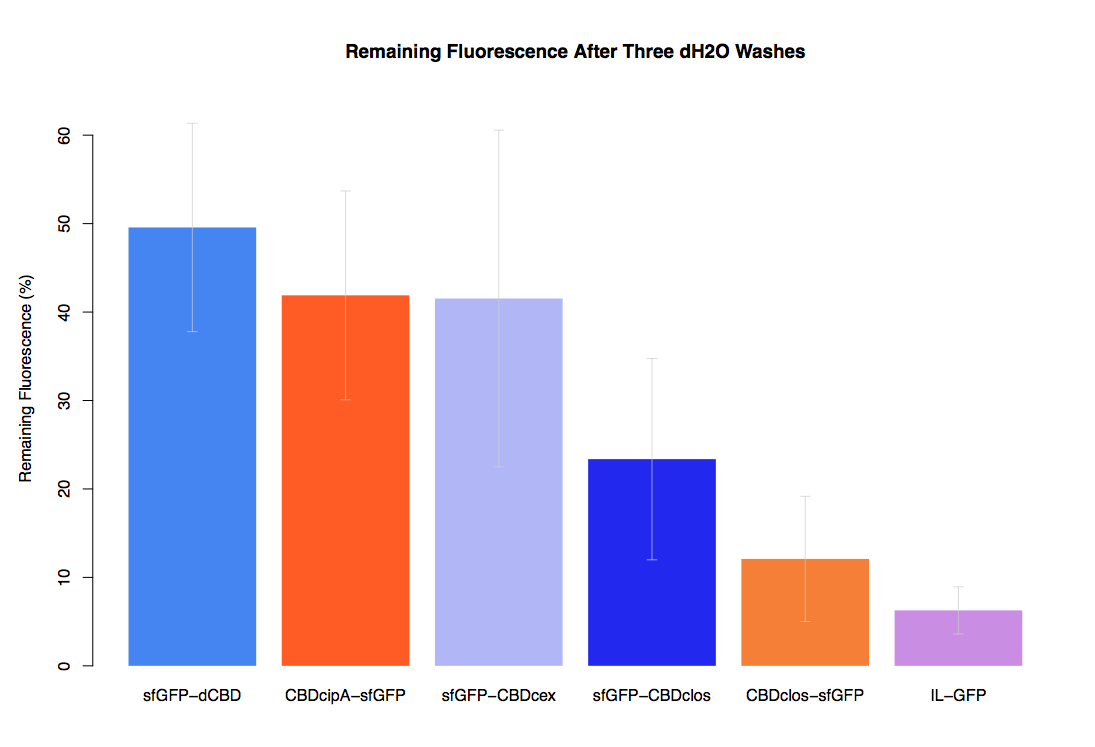
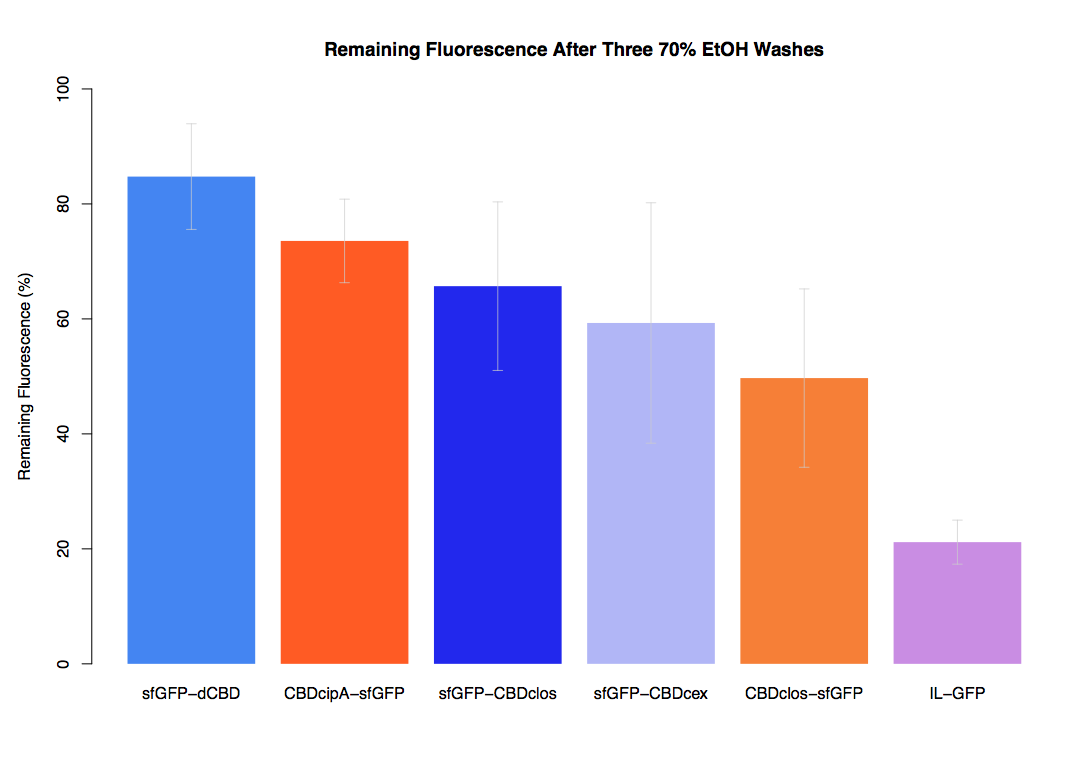
| + | In our first assay performed to determine the relative strengths of various CBDs’ binding to bacterial cellulose – represented by the percentage fluorescence left from CBDs fused to ([https://parts.igem.org/Part:BBa_K1321337 sfGFP (RFC25)]) bound to bacterial cellulose discs, when subjected to various washes - it was determined that the fusion with dCBD had the greatest binding ability in comparison to four other CBDs fused to sfGFP after three washes with both dH2O and 70% EtOH. | ||
| + | |||
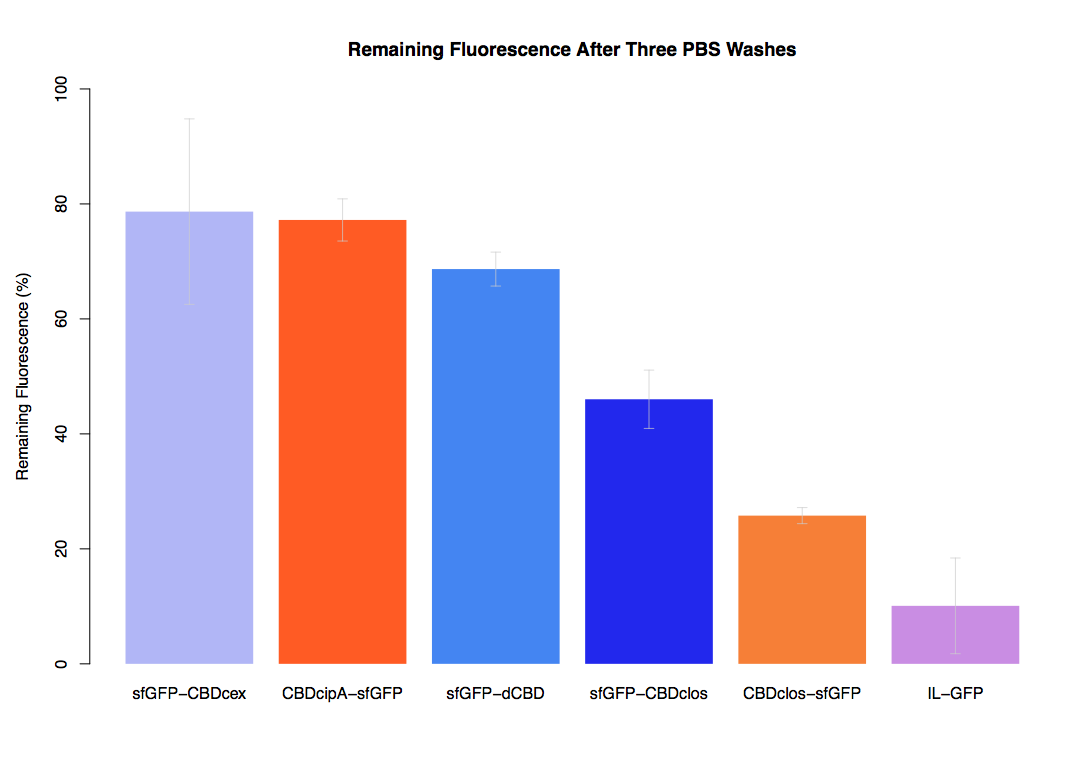
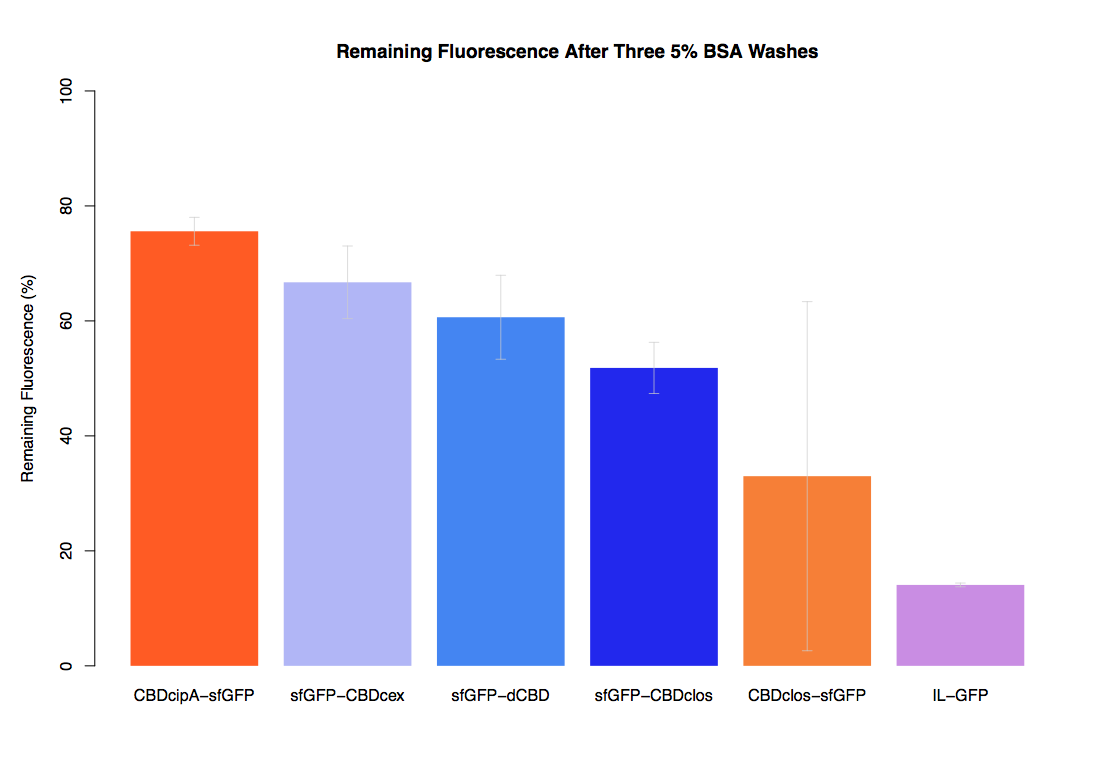
| + | In the same assay, results suggested that dCBD had on average the third greatest ability to bind bacterial cellulose, after three washes with both PBS and 5% BSA. | ||
| + | |||
| + | [[File:IC14_-_dH2Obplot1.png|700px|left|]] | ||
| + | |||
| + | [[File:IC14-EtOHbplot1.png|700px|left|]] | ||
| + | |||
| + | [[File:IC14-PBSbplot1.png|700px|left|]] | ||
| + | |||
| + | [[File:IC14-BSAbplot1.png|700px|left|]] | ||
| + | |||
| + | |||
<!-- Add more about the biology of this part here | <!-- Add more about the biology of this part here | ||
| Line 9: | Line 31: | ||
<!-- --> | <!-- --> | ||
| − | |||
<partinfo>BBa_K1321359 SequenceAndFeatures</partinfo> | <partinfo>BBa_K1321359 SequenceAndFeatures</partinfo> | ||
Latest revision as of 06:06, 2 November 2014
sfGFP fused to dCBD driven by LacI
A LacI-promoter expression construct of super-folder GFP fused N-terminally to dCBD (a cellulose-binding domain), which contains an endogenous N-terminal linker sequence.
This construct is part of a library of Super-folder GFP fusions with cellulose binding domains, which we used to assay the CBD binding affinity. Please see our [http://2014.igem.org/Team:Imperial/Functionalisation project page] for more information. The collection of sfGFP-CBD fusion parts can be seen in the table below: 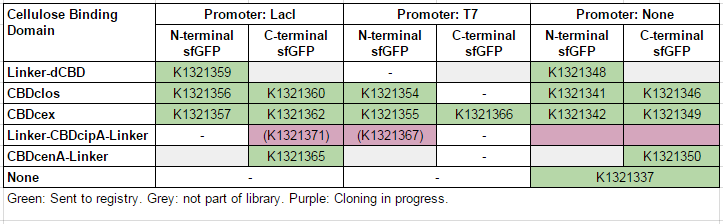
Note that the stop codon plus 6 bp at the end of the sequence are included the RFC25 suffix which is not shown. The prefix to this part is RFC10 format.
As part of our project we carried out an assay to determine the relative binding ability of CBD-sfGFP fusions to bacterial cellulose.
In our first assay performed to determine the relative strengths of various CBDs’ binding to bacterial cellulose – represented by the percentage fluorescence left from CBDs fused to (sfGFP (RFC25)) bound to bacterial cellulose discs, when subjected to various washes - it was determined that the fusion with dCBD had the greatest binding ability in comparison to four other CBDs fused to sfGFP after three washes with both dH2O and 70% EtOH.
In the same assay, results suggested that dCBD had on average the third greatest ability to bind bacterial cellulose, after three washes with both PBS and 5% BSA.
- 10COMPATIBLE WITH RFC[10]
- 12COMPATIBLE WITH RFC[12]
- 21COMPATIBLE WITH RFC[21]
- 23COMPATIBLE WITH RFC[23]
- 25INCOMPATIBLE WITH RFC[25]Illegal NgoMIV site found at 230
- 1000INCOMPATIBLE WITH RFC[1000]Illegal SapI.rc site found at 245