Difference between revisions of "Part:BBa K584000"
(→Usage and Biology) |
(→Usage and Biology) |
||
| Line 43: | Line 43: | ||
- 2% agarose gel<br> | - 2% agarose gel<br> | ||
- Quick-Load Purple 1kb Plus DNA Ladder | - Quick-Load Purple 1kb Plus DNA Ladder | ||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
<!-- --> | <!-- --> | ||
| Line 64: | Line 53: | ||
<partinfo>BBa_K584000 parameters</partinfo> | <partinfo>BBa_K584000 parameters</partinfo> | ||
<!-- --> | <!-- --> | ||
| + | |||
| + | |||
| + | <h2> <b> Northwestern 2019 - Characterization </b> </h2> | ||
| + | |||
| + | The part that we chose to characterize was BBa_K584000 a pBad GFP Generator. We wanted to characterize the optimal concentration of arabinose to induce transcription activity of pBad promoter by measuring the fluorescence per absorbance OD of our cells which have reporter gene (GFP) downstream. To perform this assay, we let E. coli bacteria containing the pBad BBa_K584000 plasmid with sfGFP grow for 9 hours in a plate reader assay. We inoculated the cultures overnight, diluted them to OD = 0.05 in the morning and we let them grow in triplicates up until OD = 0.1. Based on a previous characterization done, we added in arabinose concentration of 0.1% to 0.5% in increments of 0.1% in order to find the concentration at which pBad promoter is most actively induced. | ||
| + | |||
| + | <br> [[File:T--Northwestern--CharacterizationAssay.jpg|600px]] <br> | ||
| + | |||
| + | <br> [[File:T--Northwestern--FoldActivation.jpg|600px]] <br> | ||
| + | |||
| + | <h4> Results </h4> | ||
| + | Through performing our characterization assay, we demonstrated the growth of fluorescence per optical density of our pBad+GFP cells over time. From our data, we were able to determine the saturation point at which the pBad promoter was maximally induced. We additionally created a fold activation and dose response curve using the cells which were grown in different concentrations of arabinose. We observed through our experiments that the pBad promoter was strongly induced as the arabinose concentration was increased. | ||
Revision as of 21:17, 19 October 2019
pBAD + GFP generator
Characterization
The [http://2011.igem.org/Team:KULeuven K.U.Leuven 2011 iGEM Team] has extensively characterized the existing Biobrick part PBad promoter(BBa_I13453).
The detailed qualitative and quantitative review has been added into the experience page of the BBa_I13453 PBad promoter biobrick.
Usage and Biology
BOKU-Vienna 2019 - Characterization
When designing our project, we planned on incorporating a viability signal that expresses GFP in case of living cells under the control of an arabinose inducible promoter (pBAD). Since the construct that we cloned contained an arabinose inducible promoter (BBa_I13453) that didn’t respond to induction despite correct sequence, we decided to test the respective composite part BBa_K584000 (pBAD + GFP generator) under different arabinose concentrations.
The composite part BBa_K584000 was transformed into competent Escherischia coli (DH10B). Two clones were picked and grown on uninduced LB-media for 1 hour at 37°C on a shaker. 1mL aliquots were transferred into eprouvettes containing 1mL LB-media with different arabinose concentrations to an end concentration of 0, 0.0001, 0.001, 0.01, 0.05, 0.1 mM arabinose. Fluorescence was measured in a plate reader after 15 hours of incubation at 37°C on a shaker together with a fluorescein standard. OD-values were measured with a spectrophotometer right before samples were transferred into the 96-well plate.

Figure 1: Fluorescence at different arabinose concentrations
We calculated the mean fluorescence for each arabinose concentration across both clones, calculated the fluorescence for OD=1 and compared their fluorescence values (see figure 1). Fluorescence did not increase significantly after induction in comparison to the control of 0.04µM fluorescein, which equals the fluorescence of constantly expressed GFP under a medium strength promoter at OD600=0.5.
To verify this data, the experiment was repeated as above, but with 8 hours incubation time and different arabinose concentrations (see figure 2). Again, as in the first experiment, no significant change in fluorescence could be measured after induction.
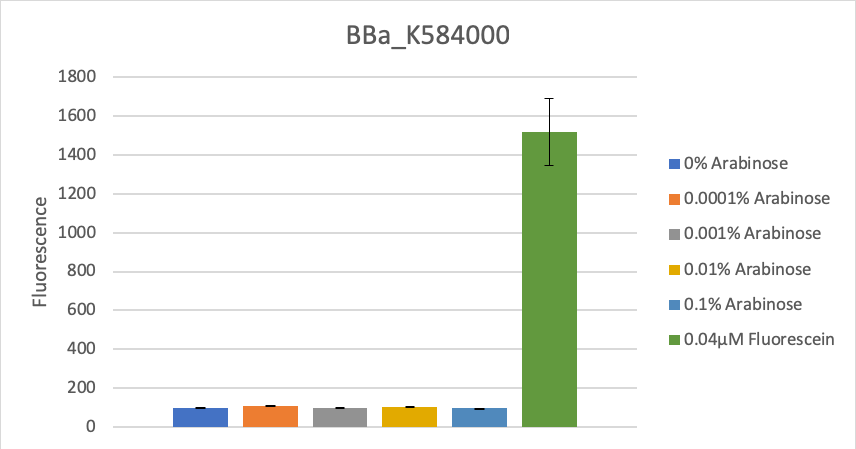
Figure 2: Fluorescence at different arabinose concentrations
Since we could not observe an induction, we checked for correctness of the plasmid by restriction digestion with the enzymes EcoRI and PstI (see figure 3).

Figure 3: Restriction digestion of BBa_K584000 - The band near 1kb matches the size of the insert, the band at 2kb the size of the empty pSB1C3 backbone
The fact that we did not observe induction of gene expression by arabinose could have different reasons. The cells might lack transporters for arabinose (e.g. araE, araFGH) or are capable of effectively degrading arabinose (e.g. by expressing araBAD genes)(Jensen et al., 1993, https://doi.org/10.1111/j.1432-1033.1993.tb19885.x).
Raw data from the plate reader here
Parameters: 485nm excitation, 525nm emission
Materials:
- LB-media with Chloramphenicol
- Tecan Infinite 200 plate reader
- white 96-well plates with white flat bottoms
- Hitachi Photometer U-1900
- L(+)-Arabinose (provider: ROTH; Art.-Nr. 5118.2)
- 2% agarose gel
- Quick-Load Purple 1kb Plus DNA Ladder
Sequence and Features
- 10COMPATIBLE WITH RFC[10]
- 12INCOMPATIBLE WITH RFC[12]Illegal NheI site found at 125
- 21INCOMPATIBLE WITH RFC[21]Illegal BamHI site found at 65
- 23COMPATIBLE WITH RFC[23]
- 25COMPATIBLE WITH RFC[25]
- 1000INCOMPATIBLE WITH RFC[1000]Illegal BsaI.rc site found at 801
Northwestern 2019 - Characterization
The part that we chose to characterize was BBa_K584000 a pBad GFP Generator. We wanted to characterize the optimal concentration of arabinose to induce transcription activity of pBad promoter by measuring the fluorescence per absorbance OD of our cells which have reporter gene (GFP) downstream. To perform this assay, we let E. coli bacteria containing the pBad BBa_K584000 plasmid with sfGFP grow for 9 hours in a plate reader assay. We inoculated the cultures overnight, diluted them to OD = 0.05 in the morning and we let them grow in triplicates up until OD = 0.1. Based on a previous characterization done, we added in arabinose concentration of 0.1% to 0.5% in increments of 0.1% in order to find the concentration at which pBad promoter is most actively induced.
Results
Through performing our characterization assay, we demonstrated the growth of fluorescence per optical density of our pBad+GFP cells over time. From our data, we were able to determine the saturation point at which the pBad promoter was maximally induced. We additionally created a fold activation and dose response curve using the cells which were grown in different concentrations of arabinose. We observed through our experiments that the pBad promoter was strongly induced as the arabinose concentration was increased.


