Difference between revisions of "Part:BBa K3198005"
Tiantian0412 (Talk | contribs) (→Results) |
Tiantian0412 (Talk | contribs) |
||
| (11 intermediate revisions by 2 users not shown) | |||
| Line 12: | Line 12: | ||
===Description=== | ===Description=== | ||
| − | This part contains a promoter that can be repressed by blue light (450nm). This promoter makes use of a blue light dependent DNA-binding protein, EL222. Irradiation by blue light of wavelength 450nm exposes the hitherto sequestered HTH, facilitating dimerization of EL222 and subsequent DNA binding. The repression is achieved by placing the DNA binding site of EL222 between the -35 and -10 hexamers of the consensus promoter in | + | This part contains a promoter that can be repressed by blue light (450nm). This promoter makes use of a blue light dependent DNA-binding protein, EL222. Irradiation by blue light of wavelength 450nm exposes the hitherto sequestered HTH, facilitating dimerization of EL222 and subsequent DNA binding. The repression is achieved by placing the DNA binding site of EL222 between the -35 and -10 hexamers of the consensus promoter in <i>Escherichia coli</i> |
| + | , creating the blue light repressible promoter PBLrep. As a result, EL222 acts as a repressor, blocking the binding of RNA polymerase and repress gene expression in the presence of blue light. In the dark, RNA polymerase can now bind, and gene expression takes place. | ||
While many teams have previously characterized blue-light repressible systems, there are not many investigations done on the potential effect of spacer DNA behind the promoter on protein expression. The part BBa K2819103 by iGEM18_NUS Singapore-A had an 11 base pair long spacer DNA behind the blue-light repressible promoter. This year, we constructed two different new plasmids. One of them had an increased spacer DNA sequence with 30 base pairs, while the other one had the spacer sequence removed completely. Our aim was to investigate how the length of this spacer DNA would affect the performance of the blue-light repressible system. | While many teams have previously characterized blue-light repressible systems, there are not many investigations done on the potential effect of spacer DNA behind the promoter on protein expression. The part BBa K2819103 by iGEM18_NUS Singapore-A had an 11 base pair long spacer DNA behind the blue-light repressible promoter. This year, we constructed two different new plasmids. One of them had an increased spacer DNA sequence with 30 base pairs, while the other one had the spacer sequence removed completely. Our aim was to investigate how the length of this spacer DNA would affect the performance of the blue-light repressible system. | ||
===Usage=== | ===Usage=== | ||
| − | This part contains a promoter that can be repressed by blue light (450nm). This promoter makes use of a blue light dependent DNA-binding protein, EL222. Irradiation by blue light of wavelength 450nm exposes the hitherto sequestered HTH, facilitating dimerization of EL222 and subsequent DNA binding. The repression is achieved by placing the DNA binding site of EL222 between the -35 and -10 hexamers of the consensus promoter in | + | This part contains a promoter that can be repressed by blue light (450nm). This promoter makes use of a blue light dependent DNA-binding protein, EL222. Irradiation by blue light of wavelength 450nm exposes the hitherto sequestered HTH, facilitating dimerization of EL222 and subsequent DNA binding. The repression is achieved by placing the DNA binding site of EL222 between the -35 and -10 hexamers of the consensus promoter in <i>Escherichia coli</i> |
| − | + | , creating the blue light repressible promoter PBLrep. As a result, EL222 acts as a repressor, blocking the binding of RNA polymerase and repress gene expression in the presence of blue light. In the dark, RNA polymerase can now bind, and gene expression takes place. | |
| − | + | ||
===Biology=== | ===Biology=== | ||
| − | Originating from the marine bacterium Erythrobacter litoralis HTCC2594, EL222 is a photosensitive DNA binding protein, with a N-terminal light-oxygen-voltage (LOV) domain and a C-terminal helix-turn-helix (HTH) DNA binding domain. | + | Originating from the marine bacterium <i>Erythrobacter litoralis</i> HTCC2594, EL222 is a photosensitive DNA binding protein, with a N-terminal light-oxygen-voltage (LOV) domain and a C-terminal helix-turn-helix (HTH) DNA binding domain. |
| − | + | ||
===Improvement over existing part [https://parts.igem.org/Part:BBa_K2819103 BBa_K2819103] by iGEM18 NUS Singapore-A=== | ===Improvement over existing part [https://parts.igem.org/Part:BBa_K2819103 BBa_K2819103] by iGEM18 NUS Singapore-A=== | ||
While many teams have previously characterized blue-light repressible systems, there are not many investigations done on the potential effect of spacer DNA behind the promoter on protein expression. The part BBa K2819103 by iGEM18_NUS Singapore-A had an 11 base pair long spacer DNA behind the blue-light repressible promoter. This year, we constructed two different new plasmids. One of them had an increased spacer DNA sequence with 30 base pairs, while the other one had the spacer sequence removed completely. Our aim was to investigate how the length of this spacer DNA would affect the performance of the blue-light repressible system. Our hypothesis is adding a longer spacer sequence would increase the RFP expression while removing the spacer sequence would decrease the RFP expression. | While many teams have previously characterized blue-light repressible systems, there are not many investigations done on the potential effect of spacer DNA behind the promoter on protein expression. The part BBa K2819103 by iGEM18_NUS Singapore-A had an 11 base pair long spacer DNA behind the blue-light repressible promoter. This year, we constructed two different new plasmids. One of them had an increased spacer DNA sequence with 30 base pairs, while the other one had the spacer sequence removed completely. Our aim was to investigate how the length of this spacer DNA would affect the performance of the blue-light repressible system. Our hypothesis is adding a longer spacer sequence would increase the RFP expression while removing the spacer sequence would decrease the RFP expression. | ||
| − | |||
| − | |||
===Characterization=== | ===Characterization=== | ||
| − | The three types of plasmid were transformed into | + | The three types of plasmid were transformed into <i>Escherichia coli</i> MG1655 strain for characterization. For simplicity, the three types of cells are referred to as 0bp, 11bp and 30bp. |
| − | 50uL of overnight culture of 0bp 11bp and 30 bp MG1655 cells were transferred into 5mL LB+Chloramphenicol medium in three 50mL tubes. They were then refreshed to a starting culture and incubated in 37 °C for one and a half hours until OD reaches about 0.8 to start characterization. | + | 50uL of overnight culture of 0bp 11bp and 30 bp MG1655 cells were transferred into 5mL LB+Chloramphenicol medium in three 50mL tubes. They were then refreshed to a starting culture and incubated in 37 °C for one and a half hours until OD<sub>600</sub> reaches about 0.8 to start characterization. |
12 well plates were used for characterization. The plate layouts are the same for both plates. | 12 well plates were used for characterization. The plate layouts are the same for both plates. | ||
| − | Plate layout: first column: triplicate of 0bp MG1655, second column: triplicate of 11bp MG1655, third column: triplicate of 30bp MG1655, last column, triplicate of blank LB medium(Figure 1). | + | Plate layout: first column: triplicate of 0bp MG1655, second column: triplicate of 11bp MG1655, third column: triplicate of 30bp MG1655, last column, triplicate of blank LB medium (Figure 1). |
| − | + | <br><br> | |
| − | https://2019.igem.org/wiki/images/9/93/T--NUS_Singapore--PartsRegistry_IP1.jpeg | + | <html> |
| − | https://2019.igem.org/wiki/images/5/5f/T--NUS_Singapore--PartsRegistry_IP2.png | + | <img style="width:400px" src="https://2019.igem.org/wiki/images/9/93/T--NUS_Singapore--PartsRegistry_IP1.jpeg"> |
| − | + | </html> | |
| − | + | <html> | |
| − | <br>< | + | <img style="width:400px" src="https://2019.igem.org/wiki/images/5/5f/T--NUS_Singapore--PartsRegistry_IP2.png"> |
| − | <br><br>1mL of each cell and medium culture were transferred into each well in the 12 well plates. Initial OD and RFP readings were taken at 0h time point using H1 Synergy microplate reader. The protocol is shaking for 10s, reading OD, and reading RFP. The results are exported as excel. For subsequent readings, the protocol is the same throughout and all results are exported in the excel sheet. | + | </html> |
| − | After the first reading, 1 plate was placed on the blue light device to be exposed to blue light, while the other plate is covered with a black cloth to prevent any exposure to light(Figure 2). Both are incubated in a shaking incubator at 37 °C and shaking at a speed of 125rpm. Hourly readings were performed for the next 8h. | + | <br><i>Figure 1: Plate layout for characterization of improved part </i> |
| − | https://2019.igem.org/wiki/images/1/15/T--NUS_Singapore--PartsRegistry_IP4.jpeg | + | <br><br>1mL of each cell and medium culture were transferred into each well in the 12 well plates. Initial OD<sub>600</sub> and RFP readings were taken at 0h time point using H1 Synergy microplate reader. The protocol is shaking for 10s, reading OD<sub>600</sub>, and reading RFP. The results are exported as excel. For subsequent readings, the protocol is the same throughout and all results are exported in the excel sheet. |
| − | < | + | After the first reading, 1 plate was placed on the blue light device to be exposed to blue light, while the other plate is covered with a black cloth to prevent any exposure to light (Figure 2). Both are incubated in a shaking incubator at 37 °C and shaking at a speed of 125rpm. Hourly readings were performed for the next 8h. |
| + | <br><br> | ||
| + | <html> | ||
| + | <img style="width:400px" src="https://2019.igem.org/wiki/images/1/15/T--NUS_Singapore--PartsRegistry_IP4.jpeg"> | ||
| + | </html> | ||
| + | <br><i>Figure 2: Experimental setup for blue light and dark conditions </i> | ||
===Results=== | ===Results=== | ||
| − | <br><br> | + | <br><br>The following figures (3 and 4) are the growth curves of cells with and without blue light exposure. |
| − | <br><br>https://2019.igem.org/wiki/images/ | + | <br><br> |
| − | < | + | <html> |
| − | <br><br>https://2019.igem.org/wiki/images/ | + | <img style="width:800px" src="https://2019.igem.org/wiki/images/e/e5/T--NUS_Singapore--PartsRegistry_003.jpeg"> |
| − | < | + | </html> |
| − | <br><br>Generally, there is no significant difference between the OD curves of cells in the dark and blue light condition. However, we noticed that under both Dark and Blue light conditions, the cells with a plasmid containing 30 base pair spacer DNA sequence grew faster and reached a higher final OD as compared to 11bp and 0bp. | + | <br><i>Figure 3: Growth curves of MG1655 transformed with plasmid containing blue-light repressible promoter and different length of spacer DNA under blue light environment</i> |
| + | <br><br> | ||
| + | <html> | ||
| + | <img style="width:800px" src="https://2019.igem.org/wiki/images/d/db/T--NUS_Singapore--PartsRegistry_004.jpeg"> | ||
| + | </html> | ||
| + | <br><i>Figure 4: Growth curves of MG1655 transformed with plasmid containing blue-light repressible promoter and different length of spacer DNA under dark condition</i> | ||
| + | <br><br>Generally, there is no significant difference between the OD<sub>600</sub> curves of cells in the dark and blue light condition. However, we noticed that under both Dark and Blue light conditions, the cells with a plasmid containing 30 base pair spacer DNA sequence grew faster and reached a higher final OD<sub>600</sub> as compared to 11bp and 0bp. | ||
| + | <br><br>The following figures (5 and 6) illustrates the production of RFP in the dark and under exposure to blue light. | ||
| + | <br><br> | ||
| + | <html> | ||
| + | <img style="width:800px" src="https://2019.igem.org/wiki/images/2/29/T--NUS_Singapore--PartsRegistry_006.jpeg"> | ||
| + | </html> | ||
| + | <br><i>Figure 5: RFP production curves of MG1655 transformed with plasmid containing blue-light repressible promoter and different length of spacer DNA under blue light environment</i> | ||
| + | <br><br> | ||
| + | <html> | ||
| + | <img style="width:800px" src="https://2019.igem.org/wiki/images/d/d6/T--NUS_Singapore--PartsRegistry_0066.jpeg"> | ||
| + | </html> | ||
| + | <br><i>Figure 6: RFP production curves of MG1655 transformed with plasmid containing blue-light repressible promoter and different length of spacer DNA under dark condition</i> | ||
| + | <br><br>We observed that the blue-light repressible system was working well. For cells incubated in the blue light environment, we could see that their RFP production decreased significantly over time while the cells incubated in the dark had an increasing RFP production. It is clear that cells containing the plasmid with 30 base pair spacer DNA had the highest RFP production overtime. The RFP production of this new construct is 3.8 fold as compared to the original construct (11 base pair). However, the cells with their spacer DNA completely removed (0 base pair) did not seem to produce RFP at all. Therefore, we can conclude that increasing the spacer DNA length behind the blue-light repressible promoter increases the RFP production while decreasing the spacer DNA reduces RFP production. | ||
| + | <br><br>Combining the findings for OD<sub>600</sub> and RFP, we had an interesting observation: The cells producing higher amount of RFP (longer spacer length) also grew faster and reached a higher final OD<sub>600</sub>. This is different from what we expected. We assumed that cells producing higher amount of RFP would have a greater metabolic burden and therefore grow slower. | ||
| + | <br><br>The following figures (7 and 8) illustrate the ratio of RFP to OD<sub>600</sub>. | ||
| + | <br><br> | ||
| + | <html> | ||
| + | <img style="width:800px" src="https://2019.igem.org/wiki/images/9/95/T--NUS_Singapore--PartsRegistry_005.jpeg"> | ||
| + | </html> | ||
| + | <br><i>Figure 7: RFP production per OD<sub>600</sub> curves of MG1655 transformed with plasmid containing blue-light repressible promoter and different length of spacer DNA under blue light environment</i> | ||
| + | <br><br> | ||
| + | <html> | ||
| + | <img style="width:800px" src="https://2019.igem.org/wiki/images/c/ca/T--NUS_Singapore--PartsRegistry_008.jpeg"> | ||
| + | </html> | ||
| + | <br><i>Figure 8: RFP production per OD<sub>600</sub> curves of MG1655 transformed with plasmid containing blue-light repressible promoter and different length of spacer DNA under dark condition</i> | ||
| + | <br><br>Taking into account of cell density, we also plotted the RFP/OD<sub>600</sub> curve. Our conclusion is clearly supported by these curves as well, with a longer spacer DNA sequence demonstrating a much larger fold change between production and repression of proteins. This therefore indicates that a larger spacer sequence would result in a more effective blue-light repressible system. | ||
| − | + | ===References=== | |
| − | + | Takakado, A., Nakasone, Y., & Terazima, M. (2017). Photoinduced dimerization of a photosensory DNA-binding protein EL222 and its LOV domain. Physical Chemistry Chemical Physics, 19(36), 24855-24865. | |
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ===Source=== | |
| − | < | + | BBa_K2819103 originated from <i>Erythrobacter litoralis</i> obtained from iGEM18_NUS_Singapore-A. |
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| + | ===Design Considerations=== | ||
| + | Generally, the basal level of protein production under blue-light repressible promoter is low. In order to better characterize the promoter and clearly show the repression, we wanted to achieve higher basal protein production. We hypothesized that a longer spacer sequence between the promoter and coding region might allow easier binding of the transcription factors and faster formation of transcription initiation complex which might lead to higher protein production. We also carefully designed the sequence of the spacer DNA such that it does not interfere with other parts of the gene, especially the coding sequence. | ||
Latest revision as of 10:41, 17 October 2019
Improved Blue light-repressible system with RFP reporter attached with YbaQ degradation tag
Team iGEM18 NUS Singapore-A submitted BBa_K2819103 which is a Blue light-repressible system with RFP reporter attached with YbaQ degradation tag.
Sequence and Features
- 10COMPATIBLE WITH RFC[10]
- 12COMPATIBLE WITH RFC[12]
- 21COMPATIBLE WITH RFC[21]
- 23COMPATIBLE WITH RFC[23]
- 25INCOMPATIBLE WITH RFC[25]Illegal AgeI site found at 641
Illegal AgeI site found at 753 - 1000COMPATIBLE WITH RFC[1000]
Description
This part contains a promoter that can be repressed by blue light (450nm). This promoter makes use of a blue light dependent DNA-binding protein, EL222. Irradiation by blue light of wavelength 450nm exposes the hitherto sequestered HTH, facilitating dimerization of EL222 and subsequent DNA binding. The repression is achieved by placing the DNA binding site of EL222 between the -35 and -10 hexamers of the consensus promoter in Escherichia coli , creating the blue light repressible promoter PBLrep. As a result, EL222 acts as a repressor, blocking the binding of RNA polymerase and repress gene expression in the presence of blue light. In the dark, RNA polymerase can now bind, and gene expression takes place.
While many teams have previously characterized blue-light repressible systems, there are not many investigations done on the potential effect of spacer DNA behind the promoter on protein expression. The part BBa K2819103 by iGEM18_NUS Singapore-A had an 11 base pair long spacer DNA behind the blue-light repressible promoter. This year, we constructed two different new plasmids. One of them had an increased spacer DNA sequence with 30 base pairs, while the other one had the spacer sequence removed completely. Our aim was to investigate how the length of this spacer DNA would affect the performance of the blue-light repressible system.
Usage
This part contains a promoter that can be repressed by blue light (450nm). This promoter makes use of a blue light dependent DNA-binding protein, EL222. Irradiation by blue light of wavelength 450nm exposes the hitherto sequestered HTH, facilitating dimerization of EL222 and subsequent DNA binding. The repression is achieved by placing the DNA binding site of EL222 between the -35 and -10 hexamers of the consensus promoter in Escherichia coli , creating the blue light repressible promoter PBLrep. As a result, EL222 acts as a repressor, blocking the binding of RNA polymerase and repress gene expression in the presence of blue light. In the dark, RNA polymerase can now bind, and gene expression takes place.
Biology
Originating from the marine bacterium Erythrobacter litoralis HTCC2594, EL222 is a photosensitive DNA binding protein, with a N-terminal light-oxygen-voltage (LOV) domain and a C-terminal helix-turn-helix (HTH) DNA binding domain.
Improvement over existing part BBa_K2819103 by iGEM18 NUS Singapore-A
While many teams have previously characterized blue-light repressible systems, there are not many investigations done on the potential effect of spacer DNA behind the promoter on protein expression. The part BBa K2819103 by iGEM18_NUS Singapore-A had an 11 base pair long spacer DNA behind the blue-light repressible promoter. This year, we constructed two different new plasmids. One of them had an increased spacer DNA sequence with 30 base pairs, while the other one had the spacer sequence removed completely. Our aim was to investigate how the length of this spacer DNA would affect the performance of the blue-light repressible system. Our hypothesis is adding a longer spacer sequence would increase the RFP expression while removing the spacer sequence would decrease the RFP expression.
Characterization
The three types of plasmid were transformed into Escherichia coli MG1655 strain for characterization. For simplicity, the three types of cells are referred to as 0bp, 11bp and 30bp. 50uL of overnight culture of 0bp 11bp and 30 bp MG1655 cells were transferred into 5mL LB+Chloramphenicol medium in three 50mL tubes. They were then refreshed to a starting culture and incubated in 37 °C for one and a half hours until OD600 reaches about 0.8 to start characterization.
12 well plates were used for characterization. The plate layouts are the same for both plates.
Plate layout: first column: triplicate of 0bp MG1655, second column: triplicate of 11bp MG1655, third column: triplicate of 30bp MG1655, last column, triplicate of blank LB medium (Figure 1).


Figure 1: Plate layout for characterization of improved part
1mL of each cell and medium culture were transferred into each well in the 12 well plates. Initial OD600 and RFP readings were taken at 0h time point using H1 Synergy microplate reader. The protocol is shaking for 10s, reading OD600, and reading RFP. The results are exported as excel. For subsequent readings, the protocol is the same throughout and all results are exported in the excel sheet.
After the first reading, 1 plate was placed on the blue light device to be exposed to blue light, while the other plate is covered with a black cloth to prevent any exposure to light (Figure 2). Both are incubated in a shaking incubator at 37 °C and shaking at a speed of 125rpm. Hourly readings were performed for the next 8h.

Figure 2: Experimental setup for blue light and dark conditions
Results
The following figures (3 and 4) are the growth curves of cells with and without blue light exposure.
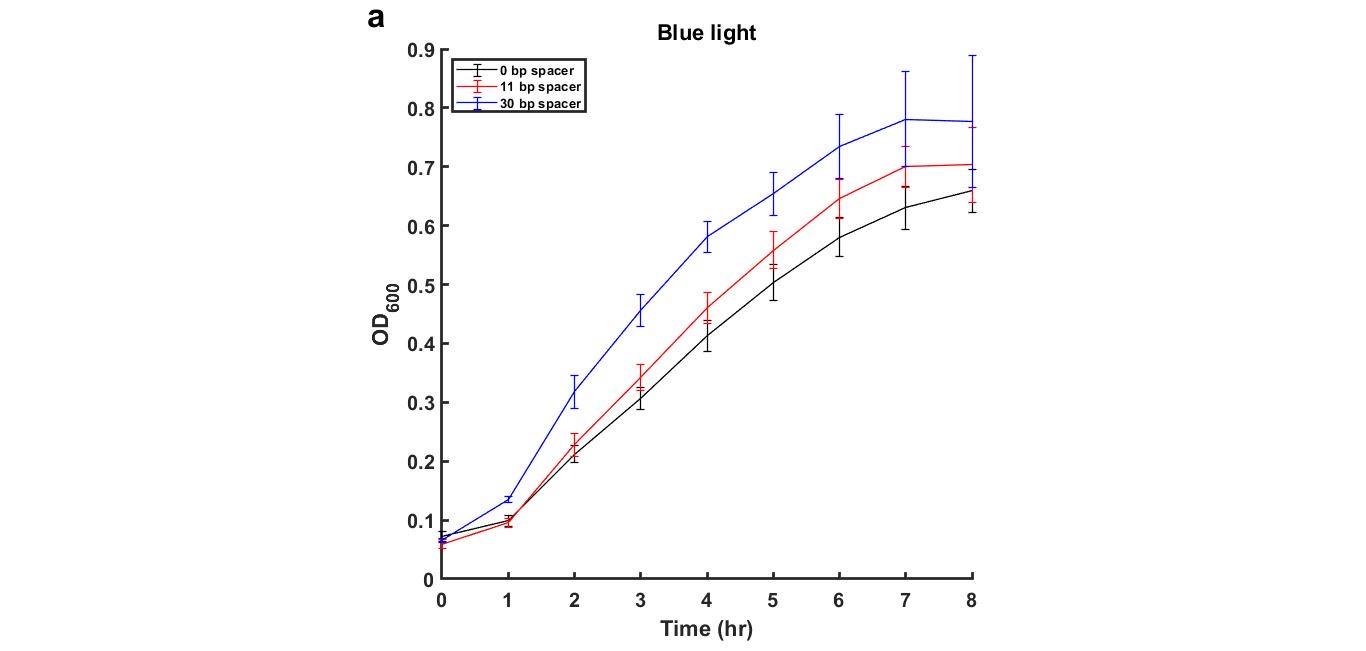
Figure 3: Growth curves of MG1655 transformed with plasmid containing blue-light repressible promoter and different length of spacer DNA under blue light environment

Figure 4: Growth curves of MG1655 transformed with plasmid containing blue-light repressible promoter and different length of spacer DNA under dark condition
Generally, there is no significant difference between the OD600 curves of cells in the dark and blue light condition. However, we noticed that under both Dark and Blue light conditions, the cells with a plasmid containing 30 base pair spacer DNA sequence grew faster and reached a higher final OD600 as compared to 11bp and 0bp.
The following figures (5 and 6) illustrates the production of RFP in the dark and under exposure to blue light.

Figure 5: RFP production curves of MG1655 transformed with plasmid containing blue-light repressible promoter and different length of spacer DNA under blue light environment

Figure 6: RFP production curves of MG1655 transformed with plasmid containing blue-light repressible promoter and different length of spacer DNA under dark condition
We observed that the blue-light repressible system was working well. For cells incubated in the blue light environment, we could see that their RFP production decreased significantly over time while the cells incubated in the dark had an increasing RFP production. It is clear that cells containing the plasmid with 30 base pair spacer DNA had the highest RFP production overtime. The RFP production of this new construct is 3.8 fold as compared to the original construct (11 base pair). However, the cells with their spacer DNA completely removed (0 base pair) did not seem to produce RFP at all. Therefore, we can conclude that increasing the spacer DNA length behind the blue-light repressible promoter increases the RFP production while decreasing the spacer DNA reduces RFP production.
Combining the findings for OD600 and RFP, we had an interesting observation: The cells producing higher amount of RFP (longer spacer length) also grew faster and reached a higher final OD600. This is different from what we expected. We assumed that cells producing higher amount of RFP would have a greater metabolic burden and therefore grow slower.
The following figures (7 and 8) illustrate the ratio of RFP to OD600.

Figure 7: RFP production per OD600 curves of MG1655 transformed with plasmid containing blue-light repressible promoter and different length of spacer DNA under blue light environment

Figure 8: RFP production per OD600 curves of MG1655 transformed with plasmid containing blue-light repressible promoter and different length of spacer DNA under dark condition
Taking into account of cell density, we also plotted the RFP/OD600 curve. Our conclusion is clearly supported by these curves as well, with a longer spacer DNA sequence demonstrating a much larger fold change between production and repression of proteins. This therefore indicates that a larger spacer sequence would result in a more effective blue-light repressible system.
References
Takakado, A., Nakasone, Y., & Terazima, M. (2017). Photoinduced dimerization of a photosensory DNA-binding protein EL222 and its LOV domain. Physical Chemistry Chemical Physics, 19(36), 24855-24865.
Source
BBa_K2819103 originated from Erythrobacter litoralis obtained from iGEM18_NUS_Singapore-A.
Design Considerations
Generally, the basal level of protein production under blue-light repressible promoter is low. In order to better characterize the promoter and clearly show the repression, we wanted to achieve higher basal protein production. We hypothesized that a longer spacer sequence between the promoter and coding region might allow easier binding of the transcription factors and faster formation of transcription initiation complex which might lead to higher protein production. We also carefully designed the sequence of the spacer DNA such that it does not interfere with other parts of the gene, especially the coding sequence.
