Difference between revisions of "Part:BBa K2571000"
Tugbainanc (Talk | contribs) |
|||
| (52 intermediate revisions by 2 users not shown) | |||
| Line 3: | Line 3: | ||
<partinfo>BBa_K2571000 short</partinfo> | <partinfo>BBa_K2571000 short</partinfo> | ||
| − | 1,2-propanediol oxidoreductase from E. coli | + | L-1,2-propanediol oxidoreductase from E. coli |
has NADH-dependent furan reductase activity. | has NADH-dependent furan reductase activity. | ||
L-1,2-propanediol oxidoreductase is an iron-dependent group III dehydrogenase | L-1,2-propanediol oxidoreductase is an iron-dependent group III dehydrogenase | ||
| − | + | ||
| − | + | ===Usage and Biology=== | |
| + | |||
FucO is the gene that codes for L-1,2-propanediol oxidoreductase which is an NADH-linked, homodimer enzyme having the role of acting on furfural which is a toxic inhibitor of microbial fermentations causing cell wall and membrane damage, DNA breaks down and reduced enzymatic activities (Zheng, 2013; Liu, Ma & Song, 2009). L-propanediol oxidoreductase is composed of two subunits with an alpha/beta Rossman nucleotide binding N-terminal domain and an all- alpha-helical C-terminal domain. (Wang et al., 2011). | FucO is the gene that codes for L-1,2-propanediol oxidoreductase which is an NADH-linked, homodimer enzyme having the role of acting on furfural which is a toxic inhibitor of microbial fermentations causing cell wall and membrane damage, DNA breaks down and reduced enzymatic activities (Zheng, 2013; Liu, Ma & Song, 2009). L-propanediol oxidoreductase is composed of two subunits with an alpha/beta Rossman nucleotide binding N-terminal domain and an all- alpha-helical C-terminal domain. (Wang et al., 2011). | ||
It belongs to iron-activated group III dehydrogenase family and also called lactaldehyde reductase (Wang et al., 2011). | It belongs to iron-activated group III dehydrogenase family and also called lactaldehyde reductase (Wang et al., 2011). | ||
| − | The enzyme catalyzes L-lactaldehyde and L-1,2- propanediol while dissimilating fucose in which acetaldehyde, ethylene glycerol, L-lactaldehyde and some more substances are used as substrates. Despite these, it takes an important role in furan reduction to its alcohol derivative (Wang et al., 2011). Moreover, the crystal structure of L-1,2-propanediol oxidoreductase is shown to be so similar with YqhD which also functions in furfural reduction (Wang et al., 2011). | + | The enzyme catalyzes L-lactaldehyde and L-1,2- propanediol while dissimilating fucose in which acetaldehyde, ethylene glycerol, L-lactaldehyde, and some more substances are used as substrates. Despite these, it takes an important role in furan reduction to its alcohol derivative (Wang et al., 2011). Moreover, the crystal structure of L-1,2-propanediol oxidoreductase is shown to be so similar with YqhD which also functions in furfural reduction (Wang et al., 2011). |
| + | |||
| + | |||
| + | |||
| + | [[File:METU_HS_Ankara FucO 3D protein structure.gif|600px|thumb|center|3D protein structure of L-1,2-propanediol oxidoreductase, METU_HS_Ankara, 2018]] | ||
| + | |||
| + | |||
| + | |||
| + | Figure represents the predicted three-dimensional structure of 1,2-propanediol oxidoreductase from <i> E. coli </i>. The protein structure of L-1,2-propanediol oxidoreductase was constructed by using Amber 14. It is demonstrated in the ribbon diagram which is done by interpolating a smooth curve through the polypeptide backbone. The colors indicate the amino acids in the protein structure. While constructing, the codon bias rule is obeyed to express the enzyme in <i> Escherichia Coli </i> KO11. | ||
| + | |||
| + | |||
| + | |||
| + | [[File:Furfural_photo.png|600px|thumb|center|Wang <i> et al </i>., 2011]] | ||
| + | |||
| + | |||
| + | |||
| + | When furfural is present in the field, the metabolism of furfural by NADPH-dependent oxidoreductases goes active in order to reduce it to its less toxic alcohol derivative - furfuryl alcohol (Zheng, 2013; Wang <i> et al. </i>, 2013; Allen <i>et al.</i> , 2010). In this metabolism, the expression of oxidoreductases that are NADPH-dependent, such as YqhD, are shown to inhibit the growth and fermentation in Escherichia coli by competing with biosynthesis for NADPH (Zheng, 2013). Therefore, the NADPH that functions as electron carriers in approximately 70 redox reactions and Pentose Phosphate Pathway’s NADPH which is an essential cofactor for self-protection against cellular stress, become poorly functional ( Allen <i>et al.</i>, 2010; Chou <i>et al.</i>, 2015). Because the native conversion of NADH to NADPH in E. coli is insufficient to revitalize the NADPH pool during fermentation, the actions shouldn’t be interfering with NADPH metabolism (Wang <i>et al</i>, 2011). Thus, the overexpression of plasmid-based NADH-dependent propanediol oxidoreductase (FucO) gene may reduce furfural to ultimately improve furfural resistance without detrimentally affecting the biosynthesis of NADPH (Wang <i>et al</i>, 2011). | ||
| + | |||
| + | |||
| + | |||
| + | |||
| + | ===Characterization=== | ||
| + | |||
| + | |||
| + | |||
| + | === Allergen Characterization: === | ||
| + | |||
| + | |||
| + | Our parts can be used in ethanol production and we used it in the lab for mass production, it was important to construct an allergenicity test. The allergenicity test makes a comparison between the sequences of the biobrick parts and the identified allergen proteins in the database. If the similarity between the biobricks and the proteins is high, it is more likely that the biobrick is allergenic. In the sliding window of 80 amino acid segments, greater than 35% means similarity to allergens. Higher similarity implies that the biobricks have a potential for negative effect to exposed populations. For more information on the protocol see the “Allergenicity Testing Protocol” in the following page http://2017.igem.org/Team:Baltimore_Bio-Crew/Experiments | ||
| + | |||
| + | Our biobrick part, BBa_K2571000 showed less than 35% match in the 80 amino acid alignments by FASTA. | ||
| + | |||
| + | |||
| + | === Gel Characterization: === | ||
| + | |||
| + | |||
| + | FucO basic part is inserted into the pSB1C3 backbone. The construct in pSB1C3 is for submission to the registry and is cultivated in DH5 alpha. | ||
| + | |||
| + | |||
| + | |||
| + | [[File:METU HS Ankara fucO basic.jpeg|600px|thumb|center| BBa_K2571000 check with FucO left and VR primers. Expected band length: 625 bp. FucO basic well show positive results.]] | ||
| + | |||
| + | |||
| + | |||
| + | FucO and VR primers are as below: | ||
| + | |||
| + | FucO left: GTGATAAGGATGCCGGAGAA | ||
| + | |||
| + | VR: ATTACCGCCTTTGAGTGAGC | ||
| + | |||
| + | |||
| + | |||
| + | === Biochemical Characterization: === | ||
| + | |||
| + | |||
| + | We inserted our basic part K2571000 which is a protein coding region in pSB1A3 backbone with a strong promoter, an RBS and a double terminator in order to test the function. | ||
| + | |||
| + | We designed our biochemical characterization experiments in order to evaluate the effects of our circuits on life span, cell mass, and ultimately the bioethanol yield of ethanologenic E. coli strain KO11. We carried out two experimental assays simultaneously. | ||
| + | |||
| + | In both of our biochemical assays, we had four cultured groups of KO11 ethanologenic strains of E.coli to test. | ||
| + | |||
| + | |||
| + | 1. KO11 un-engineered, | ||
| + | 2. KO11 with only FucO, | ||
| + | 3. KO11 with only GSH, | ||
| + | 4. KO11 with Bio-E (both FucO and GSH). | ||
| + | |||
| + | |||
| + | |||
| + | |||
| + | <b> First Assay: </b> | ||
| + | |||
| + | |||
| + | |||
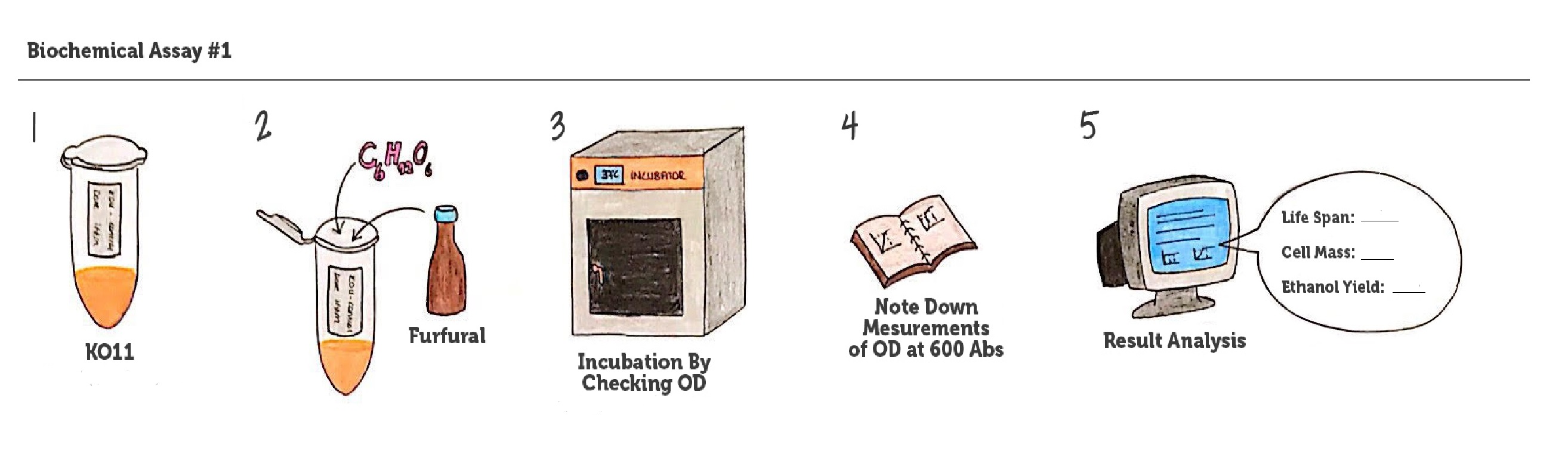
| + | [[File: METU_HS_Ankara_biochemical_assay.jpg|600px|thumb|center| Representation of our biochemical assay #1. ]] | ||
| + | |||
| + | |||
| + | |||
| + | Throughout our first assay, each group was grown in LB broth mediums containing 2% glucose and antibiotics. | ||
| + | |||
| + | To culture group #1 (KO11 un-engineered), we only added Chloramphenicol at a final concentration of 40 µg/mL since KO11 un-engineered only had resistance to Chloramphenicol in its genome; and to the mediums of the groups numbered 2, 3 and 4; we added Chloramphenicol at a final concentration of 40 µg/ml and 100 µg/ml Ampicillin. The reason was; groups 2, 3 and 4 had plasmids which carried Ampicillin resistance due to their backbone (pSB1A3). Thus, with the addition of antibiotics to the mediums, selectivity was assured. | ||
| + | |||
| + | Cultures were grown overnight, and refreshed in the morning as two sets (First set: 10 mM furfural, 2nd set: 20 mM furfural). After approximately two hours of incubation for both of the sets’ falcon groups (when they reached OD 0,6), furfural was added to their mediums. | ||
| + | |||
| + | |||
| + | <b> First Set (10 mM furfural): </b> | ||
| + | |||
| + | |||
| + | |||
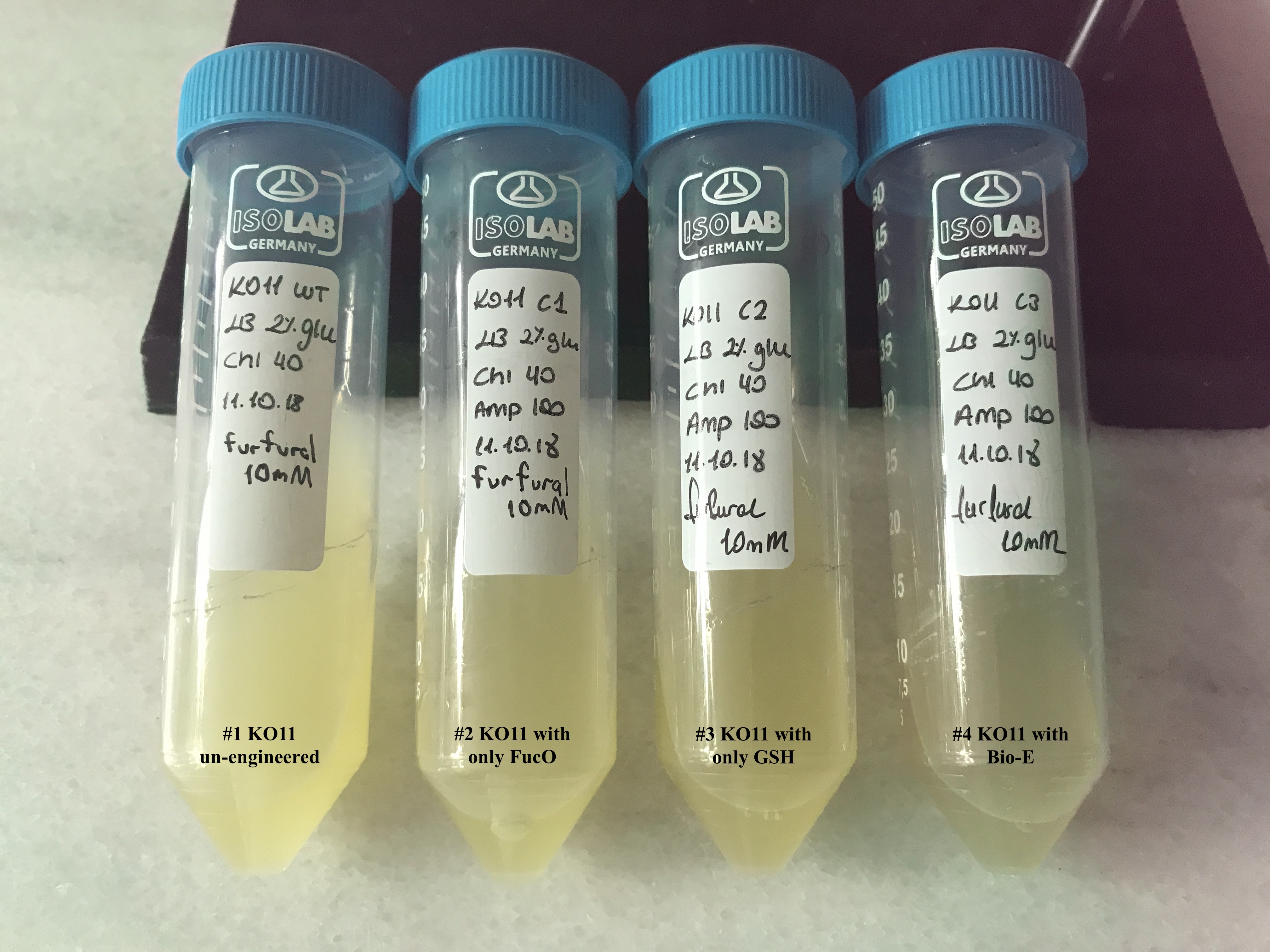
| + | [[File: T--METU_HS_Ankara_results_fig20.jpg|600px|thumb|center| Falcon tubes containing the groups for our first assay, set #1.]] | ||
| + | |||
| + | |||
| + | |||
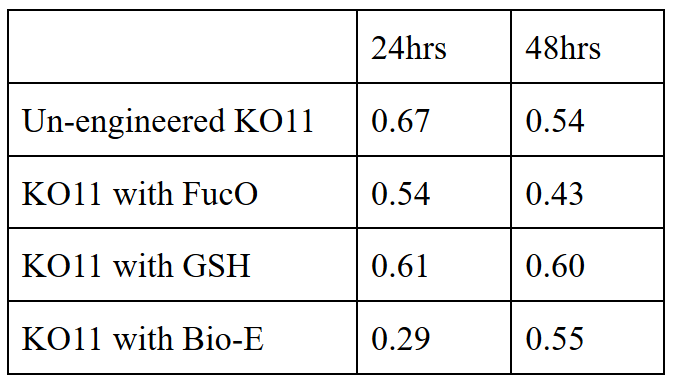
| + | We added furfural at a final concentration of 10 mM to the first four test groups’ mediums | ||
| + | and took OD measurements at Abs 600 nm with 1/10 dilution in 24 hour time intervals. | ||
| + | |||
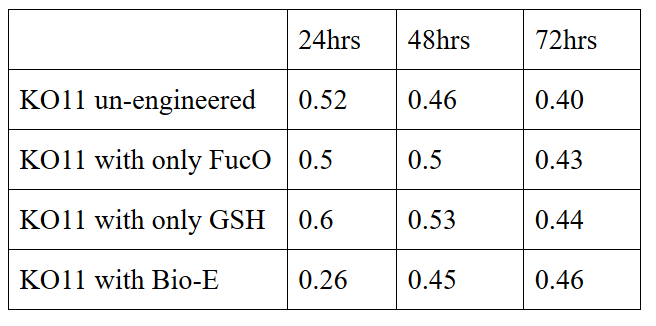
| + | 10 mM furfural OD measurements: Abs 600 nm (1/10 dilution): | ||
| + | |||
| + | |||
| + | |||
| + | [[File:METU_HS_Ankara_biochemical_assay_tablo.jpg|500px|center|]] | ||
| + | |||
| + | |||
| + | |||
| + | |||
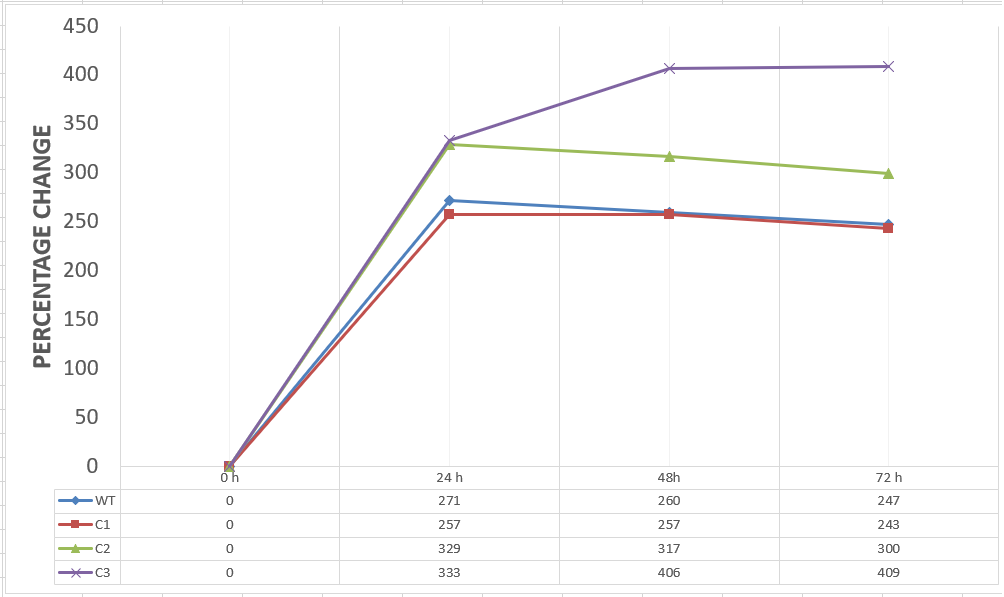
| + | [[File:T--METU HS Ankara assay 1 percentage.jpg|600px|thumb|center| Graph showing changes in cell mass of assay groups by percentage with respect to the time.]] | ||
| + | |||
| + | |||
| + | |||
| + | <b>Analysis of data:</b> | ||
| + | |||
| + | Our data demonstrated that group #1 (un-engineered) had a decrease in cell mass throughout the time verifying the inhibition of cell growth in the presence of furfural in the field. Group #2 (KO11 with only FucO) obviously gave better results with respect to un-engineered KO11. However, although the cell mass of the KO11 group with only FucO was stable in the first 48 hours, it was decreased after the 48th hour. This proves that only the presence of the gene FucO in the bacteria wasn’t enough to avoid cell mass decrease in the long term and is in need of another gene for increased tolerance. Also, only GSH’s presence isn’t enough since the group of KO11 with only GSH experienced decrease in cell mass. Group #4 (KO11 with Bio-E (both FucO and GSH)) gave measurement results as we hypothesized by continuing cellular growth in the first 48 hours and maintaining it even after the 48th hour, though at a lower rate. | ||
| + | |||
| + | |||
| + | |||
| + | <b> Second Set (20 mM furfural): </b> | ||
| + | |||
| + | |||
| + | |||
| + | [[File:T--METU_HS_Ankara_results_fig22.jpg|500px|thumb|center| Falcon tubes containing the groups for our first assay, set #2.]] | ||
| + | |||
| + | |||
| + | |||
| + | |||
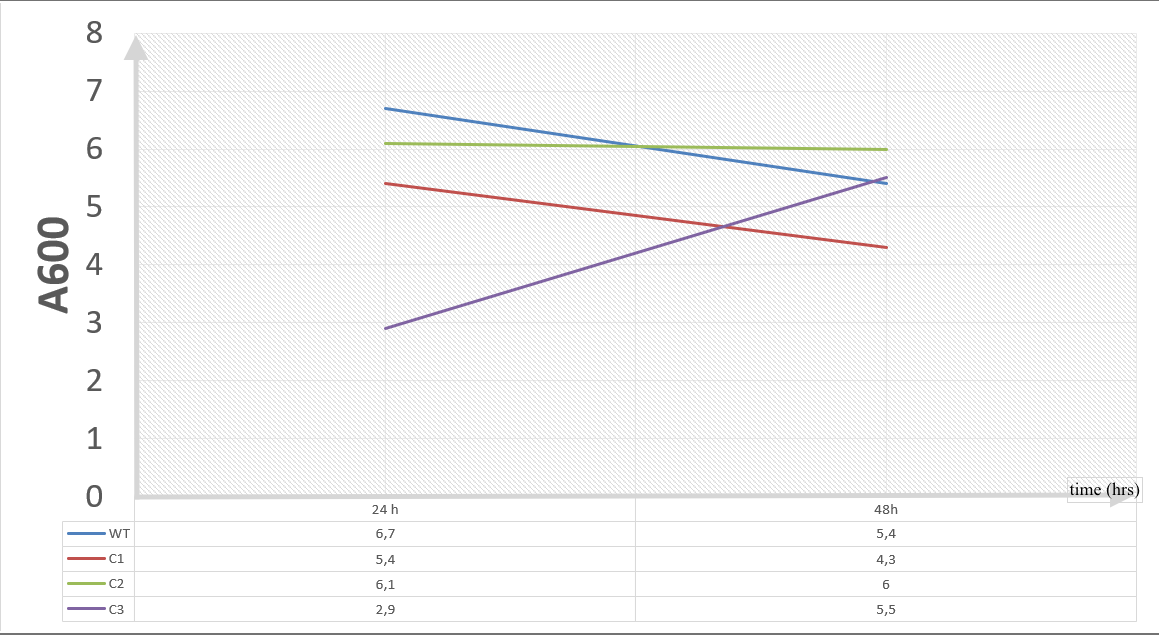
| + | To gather more information to prove our hypothesis, we designed our second experimental set and added furfural at a final concentration of 20 mM to the four test groups’ mediums followed by OD measurements at Abs 600 nm with 1/10 dilution in 24 hour time intervals. | ||
| + | |||
| + | 20 mM furfural OD measurements: Abs 600 (1/10 dilution): | ||
| + | |||
| + | |||
| + | |||
| + | |||
| + | [[File:T--METU HS Ankara assay 2 table.jpg|400px|center|]] | ||
| + | |||
| + | |||
| + | |||
| + | For our second set, we could only obtain the measurements of the first 48 hours since we faced contamination in the last day of wet-lab and we had no more time. Thus, we modelled our second experimental set’s results by demonstrating the comparison of OD results at absorbance 600. | ||
| + | |||
| + | |||
| + | |||
| + | [[File:T--METU_HS_Ankara_results_fig23.jpg|600px|thumb|center| Graph showing change in OD of assay groups with respect to the time.]] | ||
| + | |||
| + | |||
| + | |||
| + | <b> Analysis of Data: </b> | ||
| + | |||
| + | Our data demonstrated that group #1 (KO11 un-engineered) had a decrease in cell mass as time passed. Group #2 (KO11 with only FucO) also experienced a decrease in cell mass. Group #3 (KO11 with only GSH) gave better results with respect to both of the groups #1 and #2 by maintaining its cell mass stable. Group #4 (KO11 with Bio-E (both FucO and GSH)) gave the most promising measurement data as we hypothesized by continuing cellular growth (almost doubling cell mass) in the first 48 hours. | ||
| + | |||
| + | |||
| + | |||
| + | <b> Second Assay: </b> | ||
| + | |||
| + | |||
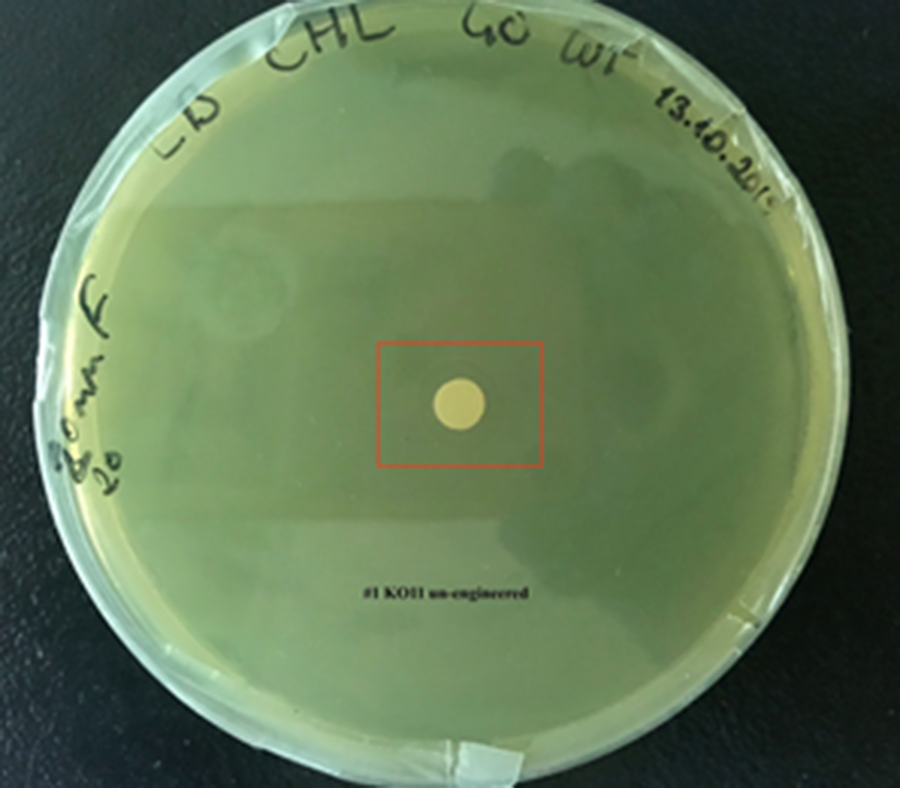
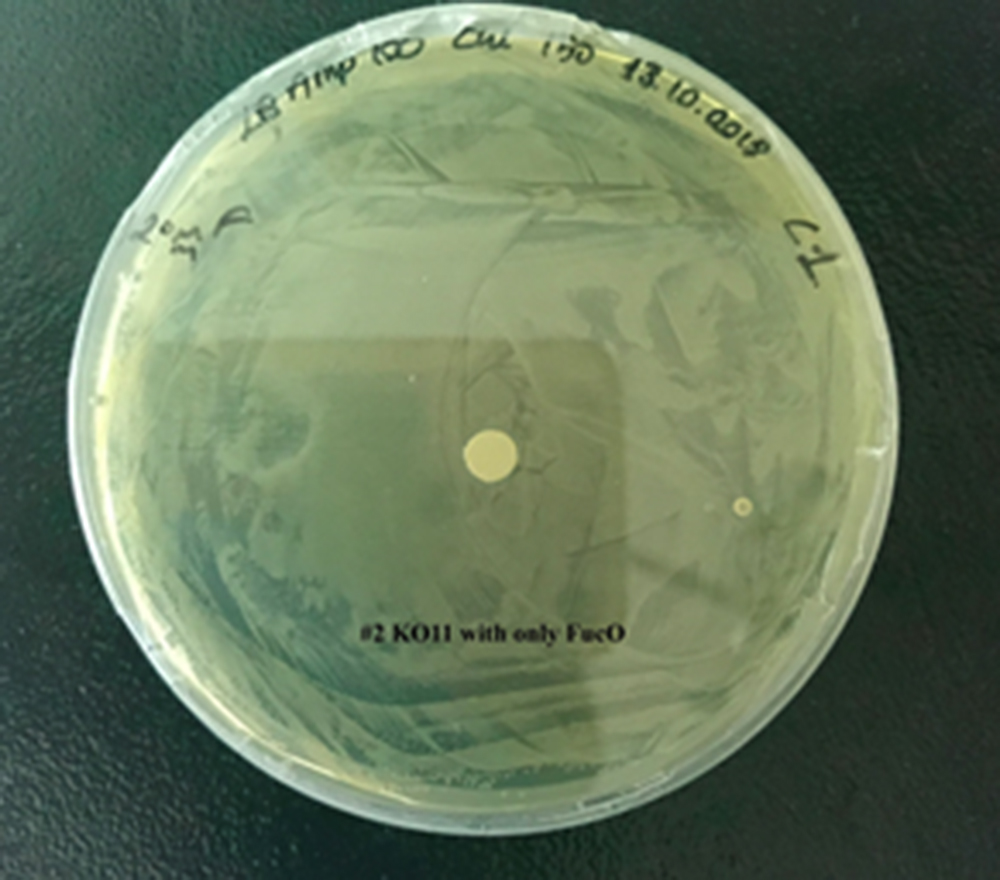
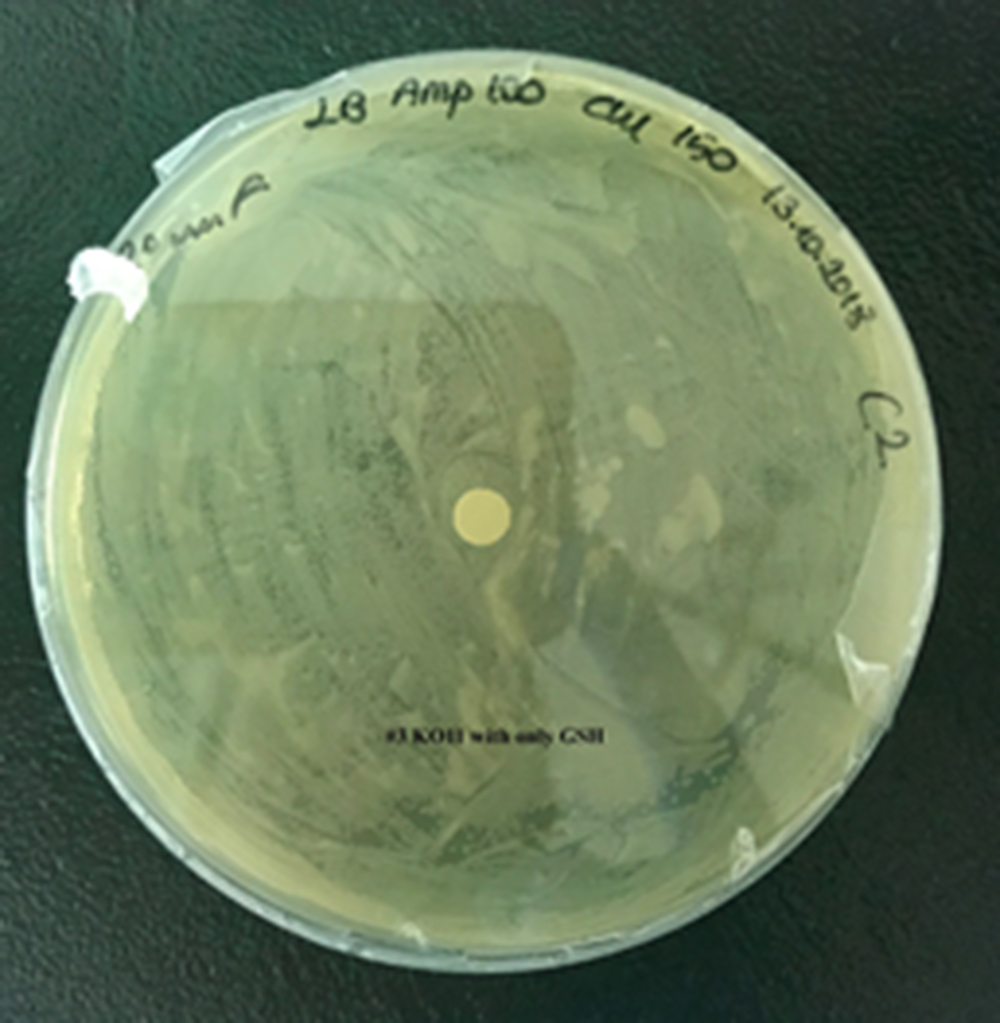
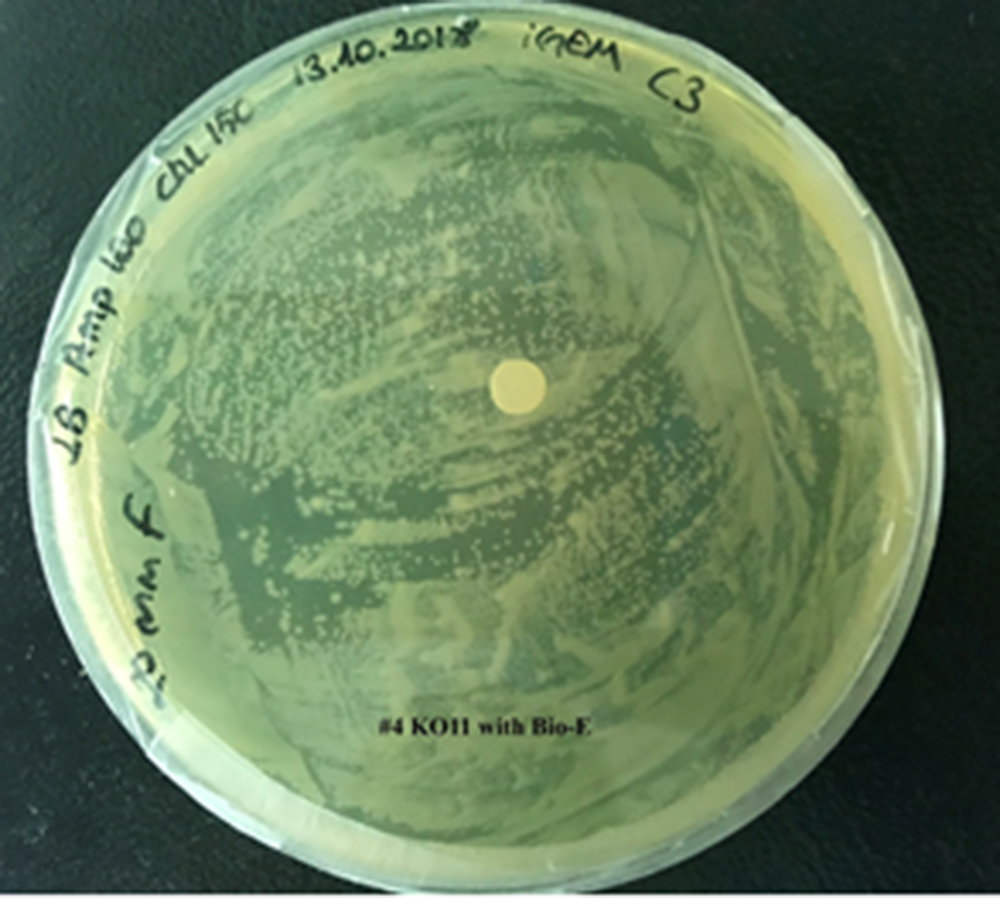
| + | For our second characterization, we followed more of qualitative evaluation to see the inhibitory zone of furfural. Firstly, we prepared a solution of furfural at a final concentration of 20 mM by diluting the stock solution with distilled H2O. Then, we soaked filter paper discs in that solution and placed them on LB agar plates hosting the four groups of our assay. | ||
| + | |||
| + | |||
| + | [[File:T--METU_HS_Ankara_results_fig24.jpg|900px|thumb|center|Image of the plates right after filter papers soaked in furfural (20 mM) were placed.]] | ||
| + | |||
| + | |||
| + | |||
| + | After 48 hours, we’ve observed a clear zone around the filter paper of the group containing KO11 un-engineered while others weren’t inhibited as much. | ||
| + | |||
| + | |||
| + | |||
| + | |||
| + | [[File:T--METU HS Ankara assay resim 2 plate.jpg|350px|thumb|left|Plate having group KO11 un-engineered. | ||
| + | ]] | ||
| + | |||
| + | |||
| + | |||
| + | [[File:T--METU HS Ankara assay resim 3 plate.jpg|350px|thumb|right|Plate having group KO11 with only FucO.]] | ||
| + | |||
| + | |||
| + | |||
| + | [[File:T--METU HS Ankara assay resim 4 plate.jpg|350px|thumb|left|Plate having group KO11 with only GSH.]] | ||
| + | |||
| + | |||
| + | |||
| + | |||
| + | [[File:T--METU HS Ankara assay resim 5 plate.jpg|350px|thumb|right|Plate having group KO11 with Bio-E (FucO and GSH.)]] | ||
| + | |||
| + | |||
| + | |||
| + | <b> | ||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | </b> | ||
| + | |||
| + | <b> Conclusion: </b> | ||
| + | |||
| + | When the quantitative measurement data and qualitative phenotypic evaluation for all of our biochemical assays are considered, we can conclude that the groups containing KO11 un-engineered are the weakest ones against furfural toxicity; and neither the group of KO11 with only FucO nor the group with only GSH is resistant enough to continue cellular growth when furfural is present in the medium. Out of four groups, only the group containing KO11 with Bio-E (both FucO and GSH) can sustain its cellular growth and survive. Overall, we can infer that our best part design (Bio-E) was successful enough to combat the inhibitive effects of furfural, indicating trueness of our hypothesis. | ||
| − | |||
| Line 27: | Line 267: | ||
<partinfo>BBa_K2571000 parameters</partinfo> | <partinfo>BBa_K2571000 parameters</partinfo> | ||
<!-- --> | <!-- --> | ||
| + | |||
===References=== | ===References=== | ||
| − | |||
| − | |||
| − | |||
| − | |||
Allen, S. A., Clark, W., McCaffery, J. M., Cai, Z., Lanctot, A., Slininger, P. J., … Gorsich, S. W. (2010). Furfural induces reactive oxygen species accumulation and cellular damage in Saccharomyces cerevisiae. Biotechnology for Biofuels, 3, 2. http://doi.org/10.1186/1754-6834-3-2 | Allen, S. A., Clark, W., McCaffery, J. M., Cai, Z., Lanctot, A., Slininger, P. J., … Gorsich, S. W. (2010). Furfural induces reactive oxygen species accumulation and cellular damage in Saccharomyces cerevisiae. Biotechnology for Biofuels, 3, 2. http://doi.org/10.1186/1754-6834-3-2 | ||
| Line 38: | Line 275: | ||
Liu, Z.L., Ma M., Song, M.(2009). Evolutionarily engineered ethanologenic yeast detoxifies lignocellulosic biomass conversion inhibitors by reprogrammed pathways. Mol Genet Genomics 282, 233-244. doi: 10.1007/s00438-009-0461-7 | Liu, Z.L., Ma M., Song, M.(2009). Evolutionarily engineered ethanologenic yeast detoxifies lignocellulosic biomass conversion inhibitors by reprogrammed pathways. Mol Genet Genomics 282, 233-244. doi: 10.1007/s00438-009-0461-7 | ||
| + | |||
| + | Wang, X., Miller, E. N., Yomano, L. P., Zhang, X., Shanmugam, K. T., & Ingram, L. O. (2011). Increased Furfural Tolerance Due to Overexpression of NADH-Dependent Oxidoreductase FucO in Escherichia coli Strains Engineered for the Production of Ethanol and Lactate. Applied and Environmental Microbiology, 77(15), 5132–5140. http://doi.org/10.1128/AEM.05008-11 | ||
Wang, X., Yomano, L. P., Lee, J. Y., York, S. W., Zheng, H., Mullinnix, M. T., … Ingram, L. O. (2013). Engineering furfural tolerance in Escherichia coli improves the fermentation of lignocellulosic sugars into renewable chemicals. Proceedings of the National Academy of Sciences of the United States of America, 110(10), 4021–4026. http://doi.org/10.1073/pnas.1217958110 | Wang, X., Yomano, L. P., Lee, J. Y., York, S. W., Zheng, H., Mullinnix, M. T., … Ingram, L. O. (2013). Engineering furfural tolerance in Escherichia coli improves the fermentation of lignocellulosic sugars into renewable chemicals. Proceedings of the National Academy of Sciences of the United States of America, 110(10), 4021–4026. http://doi.org/10.1073/pnas.1217958110 | ||
| + | |||
| + | |||
| + | Zheng, H., Wang, X., Yomano, L.P., Geddes, R. D, Shanmugan, K. T., Ingram, L.O. (2013). Improving Escherichia coli FucO for Furfural Tolerance by Saturation Mutagenesis of Individual Amino Acid Positions. Applied and Environmental Microbiology Vol 79, no 10. 3202–3208. http://aem.asm.org/content/79/10/3202.full.pdf+html | ||
Latest revision as of 19:49, 17 October 2018
FucO/ L-1,2-Propanediol Oxidoreductase
L-1,2-propanediol oxidoreductase from E. coli has NADH-dependent furan reductase activity. L-1,2-propanediol oxidoreductase is an iron-dependent group III dehydrogenase
Usage and Biology
FucO is the gene that codes for L-1,2-propanediol oxidoreductase which is an NADH-linked, homodimer enzyme having the role of acting on furfural which is a toxic inhibitor of microbial fermentations causing cell wall and membrane damage, DNA breaks down and reduced enzymatic activities (Zheng, 2013; Liu, Ma & Song, 2009). L-propanediol oxidoreductase is composed of two subunits with an alpha/beta Rossman nucleotide binding N-terminal domain and an all- alpha-helical C-terminal domain. (Wang et al., 2011). It belongs to iron-activated group III dehydrogenase family and also called lactaldehyde reductase (Wang et al., 2011).
The enzyme catalyzes L-lactaldehyde and L-1,2- propanediol while dissimilating fucose in which acetaldehyde, ethylene glycerol, L-lactaldehyde, and some more substances are used as substrates. Despite these, it takes an important role in furan reduction to its alcohol derivative (Wang et al., 2011). Moreover, the crystal structure of L-1,2-propanediol oxidoreductase is shown to be so similar with YqhD which also functions in furfural reduction (Wang et al., 2011).
Figure represents the predicted three-dimensional structure of 1,2-propanediol oxidoreductase from E. coli . The protein structure of L-1,2-propanediol oxidoreductase was constructed by using Amber 14. It is demonstrated in the ribbon diagram which is done by interpolating a smooth curve through the polypeptide backbone. The colors indicate the amino acids in the protein structure. While constructing, the codon bias rule is obeyed to express the enzyme in Escherichia Coli KO11.
When furfural is present in the field, the metabolism of furfural by NADPH-dependent oxidoreductases goes active in order to reduce it to its less toxic alcohol derivative - furfuryl alcohol (Zheng, 2013; Wang et al. , 2013; Allen et al. , 2010). In this metabolism, the expression of oxidoreductases that are NADPH-dependent, such as YqhD, are shown to inhibit the growth and fermentation in Escherichia coli by competing with biosynthesis for NADPH (Zheng, 2013). Therefore, the NADPH that functions as electron carriers in approximately 70 redox reactions and Pentose Phosphate Pathway’s NADPH which is an essential cofactor for self-protection against cellular stress, become poorly functional ( Allen et al., 2010; Chou et al., 2015). Because the native conversion of NADH to NADPH in E. coli is insufficient to revitalize the NADPH pool during fermentation, the actions shouldn’t be interfering with NADPH metabolism (Wang et al, 2011). Thus, the overexpression of plasmid-based NADH-dependent propanediol oxidoreductase (FucO) gene may reduce furfural to ultimately improve furfural resistance without detrimentally affecting the biosynthesis of NADPH (Wang et al, 2011).
Characterization
Allergen Characterization:
Our parts can be used in ethanol production and we used it in the lab for mass production, it was important to construct an allergenicity test. The allergenicity test makes a comparison between the sequences of the biobrick parts and the identified allergen proteins in the database. If the similarity between the biobricks and the proteins is high, it is more likely that the biobrick is allergenic. In the sliding window of 80 amino acid segments, greater than 35% means similarity to allergens. Higher similarity implies that the biobricks have a potential for negative effect to exposed populations. For more information on the protocol see the “Allergenicity Testing Protocol” in the following page http://2017.igem.org/Team:Baltimore_Bio-Crew/Experiments
Our biobrick part, BBa_K2571000 showed less than 35% match in the 80 amino acid alignments by FASTA.
Gel Characterization:
FucO basic part is inserted into the pSB1C3 backbone. The construct in pSB1C3 is for submission to the registry and is cultivated in DH5 alpha.
FucO and VR primers are as below:
FucO left: GTGATAAGGATGCCGGAGAA
VR: ATTACCGCCTTTGAGTGAGC
Biochemical Characterization:
We inserted our basic part K2571000 which is a protein coding region in pSB1A3 backbone with a strong promoter, an RBS and a double terminator in order to test the function.
We designed our biochemical characterization experiments in order to evaluate the effects of our circuits on life span, cell mass, and ultimately the bioethanol yield of ethanologenic E. coli strain KO11. We carried out two experimental assays simultaneously.
In both of our biochemical assays, we had four cultured groups of KO11 ethanologenic strains of E.coli to test.
1. KO11 un-engineered,
2. KO11 with only FucO,
3. KO11 with only GSH,
4. KO11 with Bio-E (both FucO and GSH).
First Assay:
Throughout our first assay, each group was grown in LB broth mediums containing 2% glucose and antibiotics.
To culture group #1 (KO11 un-engineered), we only added Chloramphenicol at a final concentration of 40 µg/mL since KO11 un-engineered only had resistance to Chloramphenicol in its genome; and to the mediums of the groups numbered 2, 3 and 4; we added Chloramphenicol at a final concentration of 40 µg/ml and 100 µg/ml Ampicillin. The reason was; groups 2, 3 and 4 had plasmids which carried Ampicillin resistance due to their backbone (pSB1A3). Thus, with the addition of antibiotics to the mediums, selectivity was assured.
Cultures were grown overnight, and refreshed in the morning as two sets (First set: 10 mM furfural, 2nd set: 20 mM furfural). After approximately two hours of incubation for both of the sets’ falcon groups (when they reached OD 0,6), furfural was added to their mediums.
First Set (10 mM furfural):
We added furfural at a final concentration of 10 mM to the first four test groups’ mediums and took OD measurements at Abs 600 nm with 1/10 dilution in 24 hour time intervals.
10 mM furfural OD measurements: Abs 600 nm (1/10 dilution):
Analysis of data:
Our data demonstrated that group #1 (un-engineered) had a decrease in cell mass throughout the time verifying the inhibition of cell growth in the presence of furfural in the field. Group #2 (KO11 with only FucO) obviously gave better results with respect to un-engineered KO11. However, although the cell mass of the KO11 group with only FucO was stable in the first 48 hours, it was decreased after the 48th hour. This proves that only the presence of the gene FucO in the bacteria wasn’t enough to avoid cell mass decrease in the long term and is in need of another gene for increased tolerance. Also, only GSH’s presence isn’t enough since the group of KO11 with only GSH experienced decrease in cell mass. Group #4 (KO11 with Bio-E (both FucO and GSH)) gave measurement results as we hypothesized by continuing cellular growth in the first 48 hours and maintaining it even after the 48th hour, though at a lower rate.
Second Set (20 mM furfural):
To gather more information to prove our hypothesis, we designed our second experimental set and added furfural at a final concentration of 20 mM to the four test groups’ mediums followed by OD measurements at Abs 600 nm with 1/10 dilution in 24 hour time intervals.
20 mM furfural OD measurements: Abs 600 (1/10 dilution):
For our second set, we could only obtain the measurements of the first 48 hours since we faced contamination in the last day of wet-lab and we had no more time. Thus, we modelled our second experimental set’s results by demonstrating the comparison of OD results at absorbance 600.
Analysis of Data:
Our data demonstrated that group #1 (KO11 un-engineered) had a decrease in cell mass as time passed. Group #2 (KO11 with only FucO) also experienced a decrease in cell mass. Group #3 (KO11 with only GSH) gave better results with respect to both of the groups #1 and #2 by maintaining its cell mass stable. Group #4 (KO11 with Bio-E (both FucO and GSH)) gave the most promising measurement data as we hypothesized by continuing cellular growth (almost doubling cell mass) in the first 48 hours.
Second Assay:
For our second characterization, we followed more of qualitative evaluation to see the inhibitory zone of furfural. Firstly, we prepared a solution of furfural at a final concentration of 20 mM by diluting the stock solution with distilled H2O. Then, we soaked filter paper discs in that solution and placed them on LB agar plates hosting the four groups of our assay.
After 48 hours, we’ve observed a clear zone around the filter paper of the group containing KO11 un-engineered while others weren’t inhibited as much.
Conclusion:
When the quantitative measurement data and qualitative phenotypic evaluation for all of our biochemical assays are considered, we can conclude that the groups containing KO11 un-engineered are the weakest ones against furfural toxicity; and neither the group of KO11 with only FucO nor the group with only GSH is resistant enough to continue cellular growth when furfural is present in the medium. Out of four groups, only the group containing KO11 with Bio-E (both FucO and GSH) can sustain its cellular growth and survive. Overall, we can infer that our best part design (Bio-E) was successful enough to combat the inhibitive effects of furfural, indicating trueness of our hypothesis.
Sequence and Features
- 10COMPATIBLE WITH RFC[10]
- 12COMPATIBLE WITH RFC[12]
- 21COMPATIBLE WITH RFC[21]
- 23COMPATIBLE WITH RFC[23]
- 25COMPATIBLE WITH RFC[25]
- 1000COMPATIBLE WITH RFC[1000]
References
Allen, S. A., Clark, W., McCaffery, J. M., Cai, Z., Lanctot, A., Slininger, P. J., … Gorsich, S. W. (2010). Furfural induces reactive oxygen species accumulation and cellular damage in Saccharomyces cerevisiae. Biotechnology for Biofuels, 3, 2. http://doi.org/10.1186/1754-6834-3-2
Chou, H.-H., Marx, C. J., & Sauer, U. (2015). Transhydrogenase Promotes the Robustness and Evolvability of E. coli Deficient in NADPH Production. PLoS Genetics, 11(2), e1005007. http://doi.org/10.1371/journal.pgen.1005007
Liu, Z.L., Ma M., Song, M.(2009). Evolutionarily engineered ethanologenic yeast detoxifies lignocellulosic biomass conversion inhibitors by reprogrammed pathways. Mol Genet Genomics 282, 233-244. doi: 10.1007/s00438-009-0461-7
Wang, X., Miller, E. N., Yomano, L. P., Zhang, X., Shanmugam, K. T., & Ingram, L. O. (2011). Increased Furfural Tolerance Due to Overexpression of NADH-Dependent Oxidoreductase FucO in Escherichia coli Strains Engineered for the Production of Ethanol and Lactate. Applied and Environmental Microbiology, 77(15), 5132–5140. http://doi.org/10.1128/AEM.05008-11
Wang, X., Yomano, L. P., Lee, J. Y., York, S. W., Zheng, H., Mullinnix, M. T., … Ingram, L. O. (2013). Engineering furfural tolerance in Escherichia coli improves the fermentation of lignocellulosic sugars into renewable chemicals. Proceedings of the National Academy of Sciences of the United States of America, 110(10), 4021–4026. http://doi.org/10.1073/pnas.1217958110
Zheng, H., Wang, X., Yomano, L.P., Geddes, R. D, Shanmugan, K. T., Ingram, L.O. (2013). Improving Escherichia coli FucO for Furfural Tolerance by Saturation Mutagenesis of Individual Amino Acid Positions. Applied and Environmental Microbiology Vol 79, no 10. 3202–3208. http://aem.asm.org/content/79/10/3202.full.pdf+html