Difference between revisions of "Part:BBa K2888010"
Yinchizhou18 (Talk | contribs) |
|||
| Line 4: | Line 4: | ||
This mlrA gene is a part of the microcystin degradation system discovered in Sphingopyxis sp. C-1. In our experiment, we intend to deal with Microcystin-LR, the most common type of microcystin toxin produced in cyanobacteria. The mlrA gene is responsible for hydrolytic cleavage and linearization of cyclic structure of MCLR. We purposely add 6x histag into our sequence in order to improve the efficacy and efficiency of expression and purification; it will also strengthen effects of binding to Ni-NTA column, making it possible to immobilize proteins and lyse MCLR repeatedly in our prototype device. | This mlrA gene is a part of the microcystin degradation system discovered in Sphingopyxis sp. C-1. In our experiment, we intend to deal with Microcystin-LR, the most common type of microcystin toxin produced in cyanobacteria. The mlrA gene is responsible for hydrolytic cleavage and linearization of cyclic structure of MCLR. We purposely add 6x histag into our sequence in order to improve the efficacy and efficiency of expression and purification; it will also strengthen effects of binding to Ni-NTA column, making it possible to immobilize proteins and lyse MCLR repeatedly in our prototype device. | ||
| + | |||
| + | |||
| + | ===Introduction=== | ||
| + | Microcystin(MC) degradation system was investigated in the MC-degrading bacteria species, Sphingopyxis sp. C-1, in previous experiment(Shimizu 2011).Among 10-more species of such bacteria, our group intends to research on the strand Sphingomonas sp, which possess a MC-degrading pathway that involves 3 genes: mrl A mrl B mrl C that works in a sequential chain reaction(Shimizu 2011) and an additional gene, mrl D, whose product is speculated to aid the transport of MCLR or its degradation process(Bourne, 2001). | ||
| + | |||
| + | https://www.omicsonline.org/publication-images/environmental-analytical-toxicology-General-chemical-8-556-g001.png | ||
| + | |||
| + | ===Our Design=== | ||
| + | |||
| + | We add 6xhistag into the coding region by PCR. Facilitating purification, 6xhistag severs as a marked purification tag for Ni-NTA column. Moreover, 6xhistag also helps expressed enzymes immobilize as well as bind to our designed prototype device. | ||
| + | <html> | ||
| + | <img src ="https://static.igem.org/mediawiki/2018/f/f3/T--SBS_SH_112144--improved_mlrA.png" width=900 height=200/> | ||
| + | <p>This BioBrick is an improvement on the one created by the Peking 2014 team (BBa_K1378001). Our composite part within prefix and suffix consists of the most commonly used promoter, RBS, 6x His tag and terminator, as well as our functional coding sequence --- mlrA gene. This composite part is assembled through Gibson Assembly; then, it is fused with linearized vector, pSB1C3 backbone, via infusion. Ultimately, we successfully extract plasmids which contain our target gene from transformed bacteria. Furthermore, we improved mlrA parts BBa_K1378001 by purposely adding 6x His tag into our sequence in order to improve the efficacy and efficiency of expression and purification; it will also strengthen effects of binding to Ni-NTA column, making it possible to immobilize proteins and lyse cyanobacteria repeatedly in our prototype device. In addition, we find that the attachment of SUMO protein can enhance the protein stability and expression system as an N-terminal fusion partner. And SUMO tag can be easily cleaved by a SUMO-specific protease in vitro during purification process.</p> | ||
| + | <img src="https://static.igem.org/mediawiki/2018/7/74/T--SBS_SH_112144--fused_mlrA.png" width=900 height300/> | ||
| + | </html> | ||
<!-- Add more about the biology of this part here | <!-- Add more about the biology of this part here | ||
Revision as of 18:29, 17 October 2018
fused mlrA gene with histag
This mlrA gene is a part of the microcystin degradation system discovered in Sphingopyxis sp. C-1. In our experiment, we intend to deal with Microcystin-LR, the most common type of microcystin toxin produced in cyanobacteria. The mlrA gene is responsible for hydrolytic cleavage and linearization of cyclic structure of MCLR. We purposely add 6x histag into our sequence in order to improve the efficacy and efficiency of expression and purification; it will also strengthen effects of binding to Ni-NTA column, making it possible to immobilize proteins and lyse MCLR repeatedly in our prototype device.
Introduction
Microcystin(MC) degradation system was investigated in the MC-degrading bacteria species, Sphingopyxis sp. C-1, in previous experiment(Shimizu 2011).Among 10-more species of such bacteria, our group intends to research on the strand Sphingomonas sp, which possess a MC-degrading pathway that involves 3 genes: mrl A mrl B mrl C that works in a sequential chain reaction(Shimizu 2011) and an additional gene, mrl D, whose product is speculated to aid the transport of MCLR or its degradation process(Bourne, 2001).
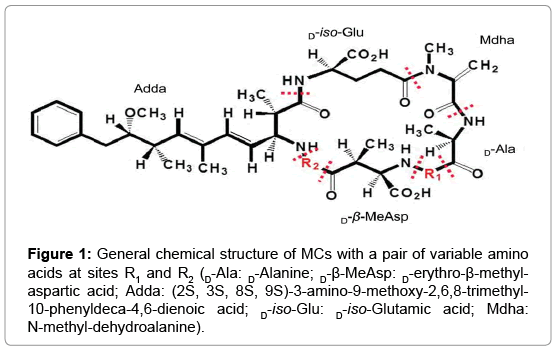
Our Design
We add 6xhistag into the coding region by PCR. Facilitating purification, 6xhistag severs as a marked purification tag for Ni-NTA column. Moreover, 6xhistag also helps expressed enzymes immobilize as well as bind to our designed prototype device.

This BioBrick is an improvement on the one created by the Peking 2014 team (BBa_K1378001). Our composite part within prefix and suffix consists of the most commonly used promoter, RBS, 6x His tag and terminator, as well as our functional coding sequence --- mlrA gene. This composite part is assembled through Gibson Assembly; then, it is fused with linearized vector, pSB1C3 backbone, via infusion. Ultimately, we successfully extract plasmids which contain our target gene from transformed bacteria. Furthermore, we improved mlrA parts BBa_K1378001 by purposely adding 6x His tag into our sequence in order to improve the efficacy and efficiency of expression and purification; it will also strengthen effects of binding to Ni-NTA column, making it possible to immobilize proteins and lyse cyanobacteria repeatedly in our prototype device. In addition, we find that the attachment of SUMO protein can enhance the protein stability and expression system as an N-terminal fusion partner. And SUMO tag can be easily cleaved by a SUMO-specific protease in vitro during purification process.

Sequence and Features
- 10COMPATIBLE WITH RFC[10]
- 12INCOMPATIBLE WITH RFC[12]Illegal NheI site found at 7
Illegal NheI site found at 30 - 21COMPATIBLE WITH RFC[21]
- 23COMPATIBLE WITH RFC[23]
- 25INCOMPATIBLE WITH RFC[25]Illegal NgoMIV site found at 770
Illegal AgeI site found at 305 - 1000COMPATIBLE WITH RFC[1000]
