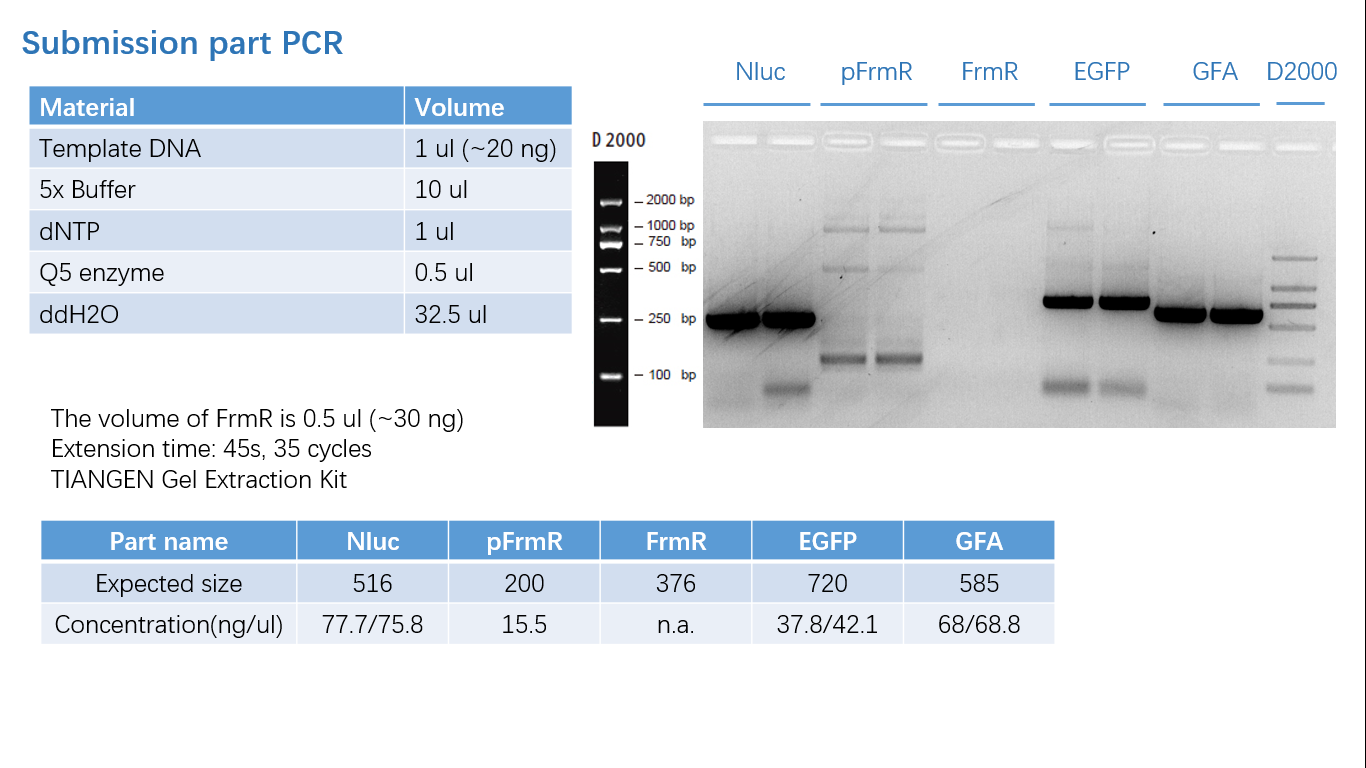
Difference between revisions of "Part:BBa K2728003"
Bluepumpkin (Talk | contribs) |
Bluepumpkin (Talk | contribs) (→Experimental Characterization) |
||
| (8 intermediate revisions by the same user not shown) | |||
| Line 24: | Line 24: | ||
=== Experimental Characterization === | === Experimental Characterization === | ||
| − | We used the | + | We used the kit provided by Promega to test its luminescence expresssion. We transferred the plasmid into E. coli BL21 strain, a common bacterial strain for protein expression. After overnight culture and adding substrate, we were able to witness the luminescence of Nanoluc in contrast of normal bacteria. |
<br /> | <br /> | ||
| + | [[File:Ep5.png|left|500px]]<br clear=all> | ||
| + | [[File:T--BGIC-Global--nanoluccorrection.jpg|left|500px]]<br clear=all> | ||
<br /> | <br /> | ||
| + | <br /> | ||
| + | |||
=== Potential Application === | === Potential Application === | ||
The nanoluc luciferase can be used as a reporter of formaldehyde. We can calculate the formaldehyde concentration according to the strength of luminescence and the model of luminescence-formaldehyde concentration. What’s more, it can be used in the product so that customers can judge the existence of formaldehyde by observing the luminescence. | The nanoluc luciferase can be used as a reporter of formaldehyde. We can calculate the formaldehyde concentration according to the strength of luminescence and the model of luminescence-formaldehyde concentration. What’s more, it can be used in the product so that customers can judge the existence of formaldehyde by observing the luminescence. | ||
Latest revision as of 03:09, 17 October 2018
NanoLuc
Basic Description
NanoLuc (Nluc) luciferase is a small enzyme (19.1kDa) engineered for optimal performance as a luminescent reporter. The enzyme is about 100-fold brighter than either firefly (Photinus pyralis) or Renilla reniformis luciferase using a novel substrate, furimazine, to produce high intensity, glow-type luminescence. The luminescent reaction is ATP-independent and designed to suppress background luminescence for maximal assay sensitivity.
Sequence and Features
NanoLuc luciferase possesses a number of physical properties that make it an excellent reporter protein:
- very small, monomeric enzyme (171 amino acids; 513bp)
- high thermal stability (Tm = 60°C)
- active over a broad pH range (pH 6–8)
- no post-translational modifications or disulfide bonds
- uniform distribution in cells
- emission spectrum well suited for bioluminescence resonance energy transfer (BRET; λmax = 465nM)
Origin
Nanoluc luciferase is a commercial product from PROMEGA. It was engineered by directed evolution from a deep-sea shrimp (Oplophorus gracilirostris) luciferase.
Experimental Characterization
We used the kit provided by Promega to test its luminescence expresssion. We transferred the plasmid into E. coli BL21 strain, a common bacterial strain for protein expression. After overnight culture and adding substrate, we were able to witness the luminescence of Nanoluc in contrast of normal bacteria.
Potential Application
The nanoluc luciferase can be used as a reporter of formaldehyde. We can calculate the formaldehyde concentration according to the strength of luminescence and the model of luminescence-formaldehyde concentration. What’s more, it can be used in the product so that customers can judge the existence of formaldehyde by observing the luminescence.
Parts Verification Before Submission
We verified our parts in the lab before submission. They are reliable! Please feel free to apply them onto your project.=)
Fig 1: PCR (to get targeted genes)
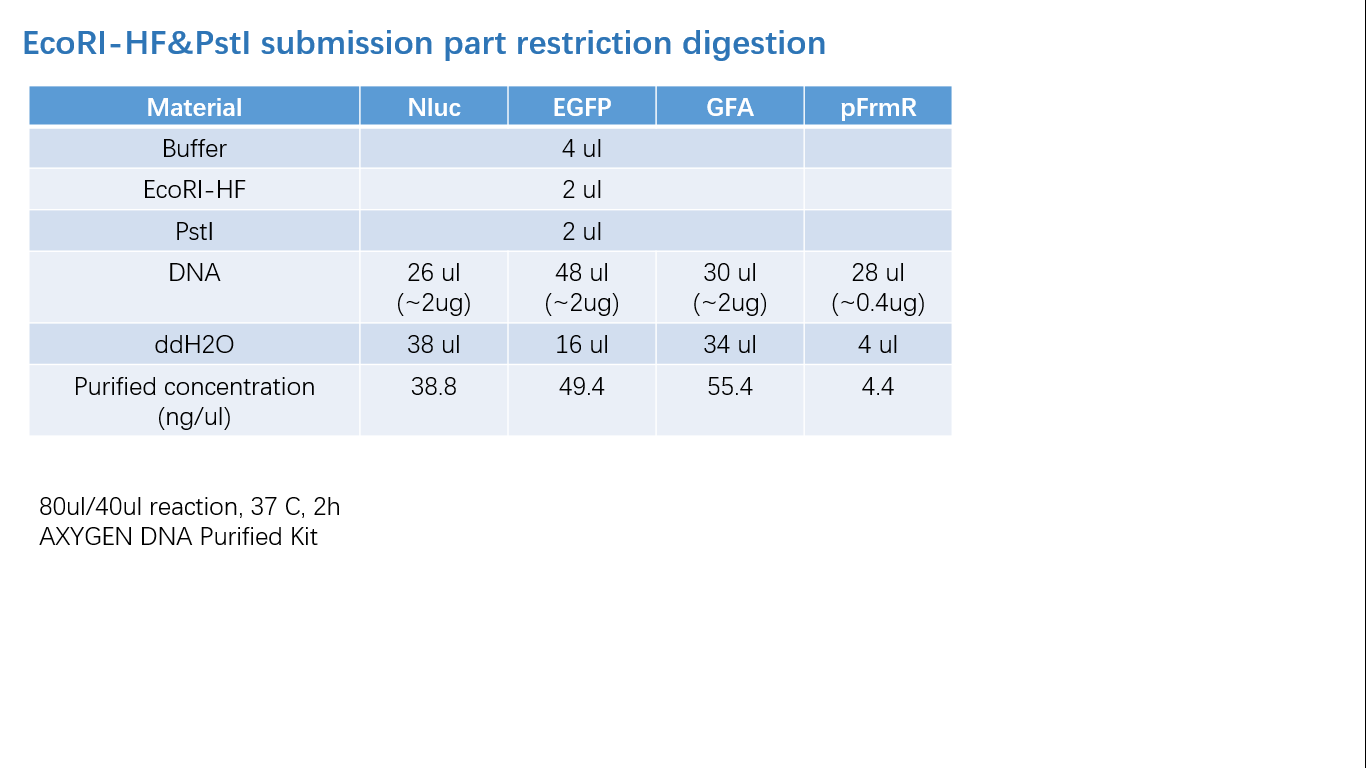
Fig 2: Restriction Digestion
Fig 3: Ligation
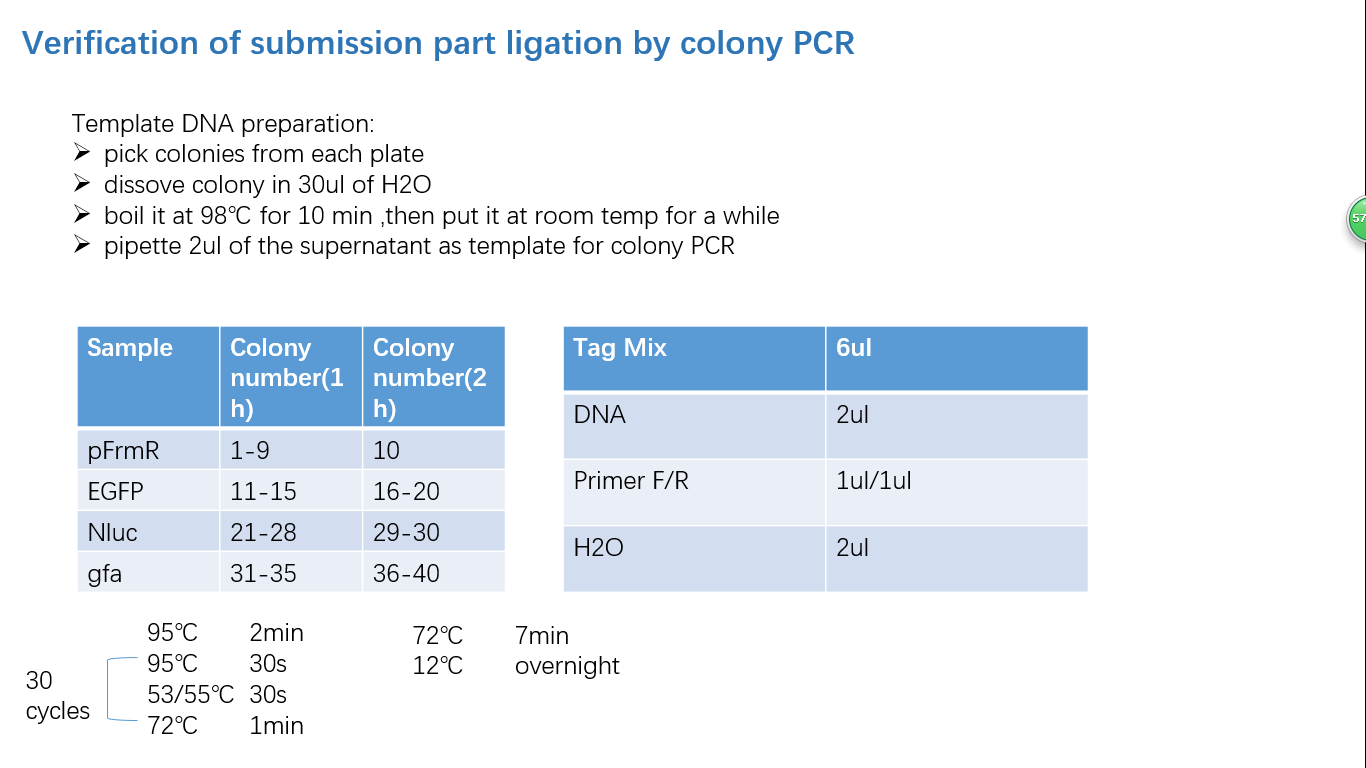
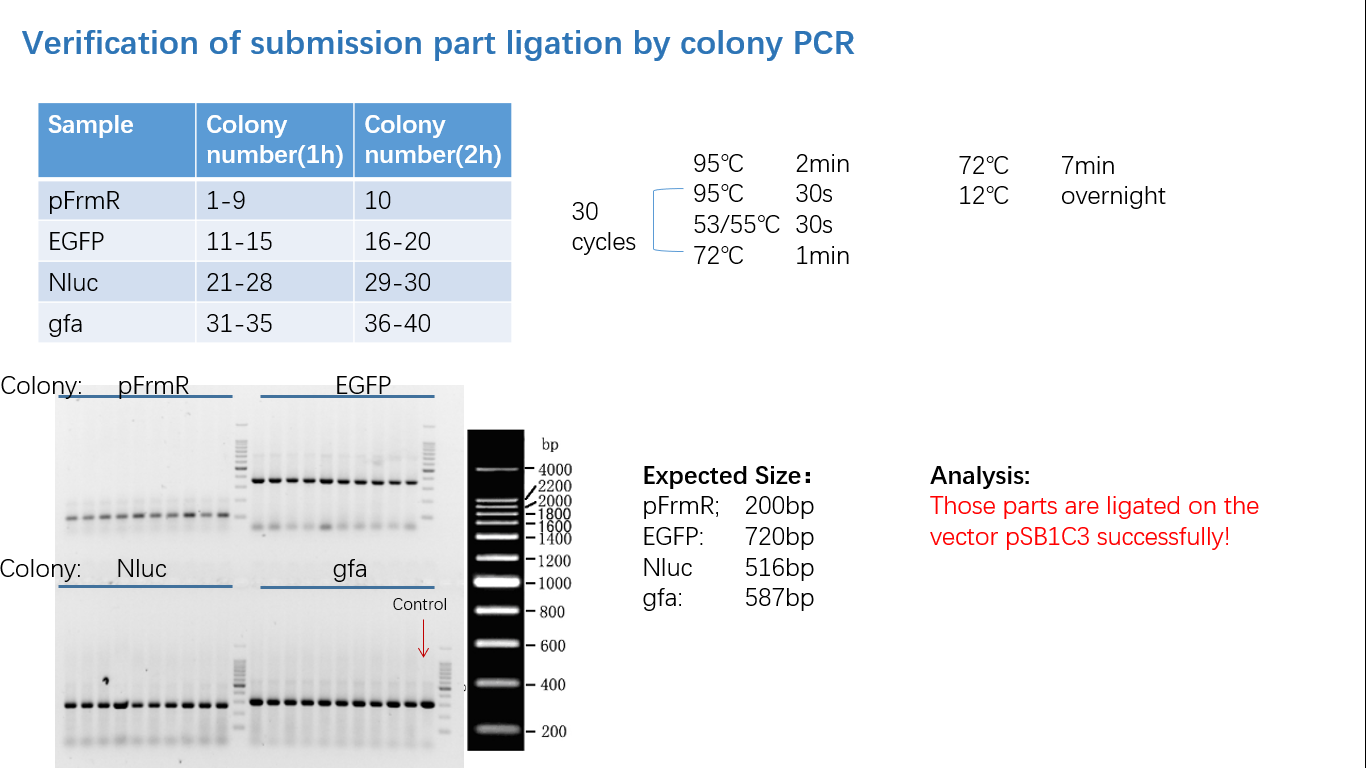
Fig 4: Colony PCR
Fig 5: Gel Verification
References
Goyet, E., et al., Fast and high resolution single-cell BRET imaging. Sci Rep, 2016. 6: p. 28231.
Sequence and Features
- 10COMPATIBLE WITH RFC[10]
- 12COMPATIBLE WITH RFC[12]
- 21COMPATIBLE WITH RFC[21]
- 23COMPATIBLE WITH RFC[23]
- 25INCOMPATIBLE WITH RFC[25]Illegal NgoMIV site found at 46
- 1000COMPATIBLE WITH RFC[1000]