Difference between revisions of "Part:BBa K2457002"
Giomaklouf (Talk | contribs) |
|||
| (17 intermediate revisions by 4 users not shown) | |||
| Line 2: | Line 2: | ||
__NOTOC__ | __NOTOC__ | ||
<partinfo>BBa_K2457002 short</partinfo> | <partinfo>BBa_K2457002 short</partinfo> | ||
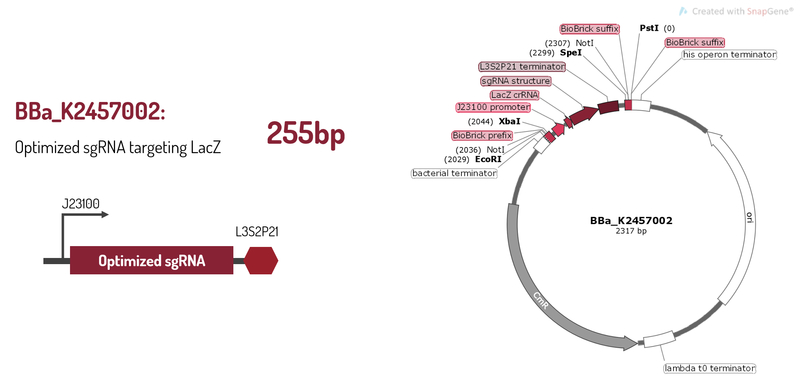
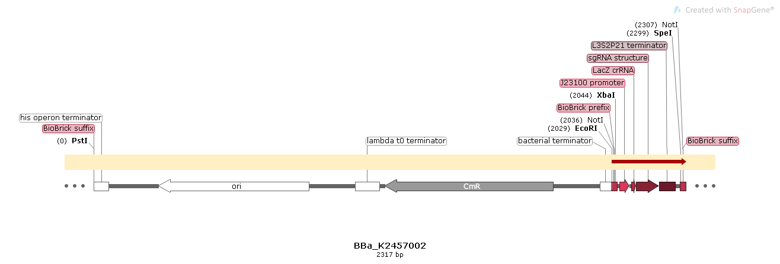
| − | BBa_K2457002 was engineered to be a building block of the standard BioBrick toolbox for bacterial genome: the ([http://2017.igem.org/Team:Amazonas_Brazil pCRISPeasy]). It’s the first BioBrick available on Registry with an improved sgRNA sequence - both crRNA design and tracrRNA secondary structure - to increase the CRISPR-Cas9 editing efficiency. BBa_K2457002 is composed by: the J23100 constitutive promoter sequence + an optimized sgRNA (crRNA with a GG motif at the 3’ end of it’s target-specific sequence from LacZ [1] and tracrRNA with extended duplex length and a mutation at the thymine 4 [2]) + L3S2P21 terminator [3]. It was rationally designed to be standardized AND interchangeable, aiming to provide a BioBrick to attend multiple genome editing purposes. For that matter, It’s easy to engineer new modules aiming to insert information into the genome through RFC10 Assembly. | + | <p>BBa_K2457002 was engineered to be a building block of the standard BioBrick toolbox for bacterial genome: the ([http://2017.igem.org/Team:Amazonas_Brazil pCRISPeasy]). It’s the first BioBrick available on Registry with an improved sgRNA sequence - both crRNA design and tracrRNA secondary structure - to increase the CRISPR-Cas9 editing efficiency. BBa_K2457002 is composed by: the J23100 constitutive promoter sequence + an optimized sgRNA (crRNA with a GG motif at the 3’ end of it’s target-specific sequence from LacZ [1] and tracrRNA with extended duplex length and a mutation at the thymine 4 [2]) + L3S2P21 terminator [3]. It was rationally designed to be standardized AND interchangeable, aiming to provide a BioBrick to attend multiple genome editing purposes. For that matter, It’s easy to engineer new modules aiming to insert information into the genome through RFC10 Assembly.</p> |
| + | |||
| + | [[File:BBa K2457002 circuit.png]] | ||
| + | |||
| + | Figure 1: BBa_K2457002 circuit. | ||
| + | |||
| + | [[File:SgRNA_ESTRUTURA.png]] | ||
| + | |||
| + | Figura 2: BBa_K2457002 structure. | ||
===Usage and Biology=== | ===Usage and Biology=== | ||
| + | <p>Single-guide RNA (sgRNA) is one of the two building blocks of the CRISPR-Cas9 genome editing machinery, which directs the Cas9 protein to introduce double-stranded breaks (DSB) into target genomes sequences.</p> | ||
| + | <p>In nature, after the memory development fase made by Cas proteins, the CRISPR locus is transcribed by RNA polymerase synthesizing the crRNA precursor.The post-transcriptional maturation process begins, when this pre-crRNA ,through the complementarity its palindromic repetition, hybridizes itself with a transactivator RNA (tracrRNA), catalyzing the double strand ribonuclease formation RNAses III with the Cas9 protein. This complex cleaves the upstream palindromic repeated hairpin, making small hybridized crRNAs with the transactivator RNA (tracrRNA) in a duo composed by crRNA-tracrRNA, carrying a specific protospacer (complementary sequence) from the invasor DNA. This system, demonstrated on Figure 2 below, Deltcheva et al suggested in 2011, having as inspiration the microRNAs by interference RNAs in eukaryotes.</p> | ||
| + | <p>Therefore, the complex crRNA-tracrRNA consists of: a crRNA, with 42 nucleotides, deriving from the pre-crRNA transcribed on locus CRISPR; and a tracrRNA with 89 nucleotides, transcribed upstream to Cas operon. This non-coding RNAs hybridize themselves by Rosalind-Watson-Crick base pairing, composing a more negatively charged structure. Through polar interaction and bases stacking, the crRNA-tracrRNA is adjusted in a positive charged site between REC and NUC lobuses interface originated at Cas9 protein stemming from Streptococcus pyogenes, changing its quaternary structure to a catalytically active ribonucleoprotein complex (RNP). It has the ability to cleave DNA target sequences that are complementary to the 20 guide nucleotides present in crRNA through the HNH and RuvC nuclease domains.</p> | ||
===Design=== | ===Design=== | ||
| + | <b>Synthetic sgRNA structures</b> | ||
| + | <p>Nowadays, the first synthetic chimera of native guide RNA, built by Jinek et al in 2012, is the RNA structure commonly applied in CRISPR Cas9-based scientific projects. This structure has a shorter duplex when compared with native (Fig. 4) composed by: I) a guide sequence with 20 bp + linker loop GAAA + 30 bp of RNA-transactivator tracrRNA.</p> | ||
| + | <p>However, although this structure widely used containing 30 bp of tracrRNA tail is the minimum region for Cas9 interaction, in order to form its ribonucleoprotein complex catalytically active (RNP) in vitro. Guide RNAs, as an extended tail (+67 till +87), drastically enhance Cas9 activity in vivo (Dang et al. 2015)</p> | ||
| + | |||
| + | ===Assembly=== | ||
| + | The sequence was ordered as gBlocks from Integrated DNA Technologies and it was assembled by restriction enzyme site. | ||
| + | |||
| + | [[File: SgRNA_Dig.jpg]] | ||
| + | |||
| + | Figure 3: Eletrophoretic profile of restriction digestion of BBa_K2457002. | ||
===Characterization=== | ===Characterization=== | ||
| + | <p>BBa_K2457002 was used by Amazonas_Brazil team in two methods based on standard BioBricks modules for bacterial genome engineering mediated by CRISPR-Cas9 machinery: the CRISPeasys. The approaches constructed were 1) for non-homologous end joining (NHEJ) repair pathway, the CRISPeasy-out method (or crOUT), referring to knock-out and the 2) for homologous directed repair (HDR), the CRISPeasy-in method (or crIN), referring to knock-in. </p> | ||
| + | |||
| + | [[File:CROUT CRIN 1.png]] | ||
| + | <p>Figure 4: CRISPeasy subdivisions.</p> | ||
| + | |||
| + | <p><b>1. CROUT (CRISPeasy-out method): Designed for knock-out strategy for bacterial genome editing based on CRISPR/Cas9</b></p> | ||
| + | <p>In order to characterize our genetic modules, It was transformed by electroporation <i>E. coli</i> K12 MG1655 with BBa_K2457006 (pBAD_Cas9_sgRNA). We took isolated colonies to pre-inoculate in 5mL of Luria-Bertani (LB), incubating overnight in a shaker at 37ºC. After that, we measured the Optical Density (OD) on spectrophotometer 600 nm aiming to standardize the cell ratio to 1 OD. All the samples were inoculated to 0.05 OD. Our experiment was performed in 96-well plates with 200µL of LB + 25µg/mL chloramphenicol, measuring the cell growth for over 8h in a range of conditions (see the layout plate below). We implemented Hidex Chameleon spectrophotometer to measure the OD and incubate the 96-well plates into shaker at 37°C. As a control, we employed MG1655 carrying two genetic circuits: BBa_K2457001 (Standardized Cas9 coding sequence without regulatory modules) AND BBa_K2457005 (Cas9 device without sgRNA).</p> | ||
| + | |||
| + | <p>Glucose 1% was applied as a repressor to understand the activation and deactivation of the Cas9 expression and was added in 50% of the samples. The L-Arabinose solution is used as the inductor of the AraC_pBAD regulatory module controlling the Cas9 expression and was added in 75% of the samples, on respective concentrations: 0,05%, 0,1% and 0,2%. It was added at 1, 2, 3 and 4 hours of cell growth.</p> | ||
| + | |||
| + | [[File:CROUT 1.png]] | ||
| + | |||
| + | [[File:CROUT 2.png]] | ||
| + | |||
| + | [[File:CROUT 3.png]] | ||
| + | <p>Figure 5: CRISPeasy-out results.</p> | ||
| + | |||
| + | <p>Based on a range of experiments involving the knocking-out process, we could propose that in the glucose (repressor) absence AND in the arabinose presence (inductor), MG1655 transformed with the BBa_K2457006 vector, carrying the Cas9 device and constitutively expressing sgRNA, reported a meaningful lower growth in contrast with the samples without the regulatory module (BBa_K2457001) OR without the sgRNA (BBa_K2457005). And, in the glucose presence, repressing the Cas9 expression, all the samples displayed a similar growth performance. The samples which carried the sgRNA (i.e., directing the Cas9 sequence to lead the DSB into a specific point of the genome) presented a low cell viability, supporting the hypothesis that even occurring the Non-Homologous End Joining (NHEJ) cell repair, not all cells could recover themselves, leading to the cellular death. Thus, we founded our proof of concept on the well-characterized impact caused by the Cas9 activity into the cellular machinery AND cell growth.</p> | ||
| + | |||
| + | <p><b>2. CRIN (CRISPeasy-in method): Designed for knock-in strategy for bacterial genome editing based on CRISPR/Cas9</b></p> | ||
| + | |||
| + | <p>As the main key to this method, there is an AND logic gate composed of Cas9 coupled with RecA, which operates the Donor DNA insertion into <i>E. coli</i> genome through Direct Homology Repair (HDR). In this way, the arabinose AND IPTG (the inputs) activate the gene expression program encoding Cas9 and RecA. Then, there is the execution of their hardware's (physical parts, e.g. proteins) operation. As an output, Cas9 AND RecA activate the DNA insertion into the genome. | ||
| + | In order to standardize a reliable datasheet of genome engineering efficiency, we tested a range of electrocompetent <i>E. coli</i> K12 MG1655 cells with different circuits: test A) carrying a non-pre-inducted Cas9 device + sgRNA (BBa_K2457006) plasmid; B) carrying a pre-inducted Cas9 AND RecA; C) carrying a pre-inducted RecA. The donor DNA was electroporated in all the tests. </p> | ||
| + | |||
| + | [[File:CRIN 2 METHODOLOGY.png]] | ||
| + | |||
| + | Figure 6: CRISPeasy-in methodology. | ||
| + | |||
| + | <p><b>A) carrying a non-pre-inducted Cas9 device + sgRNA;</b></p> | ||
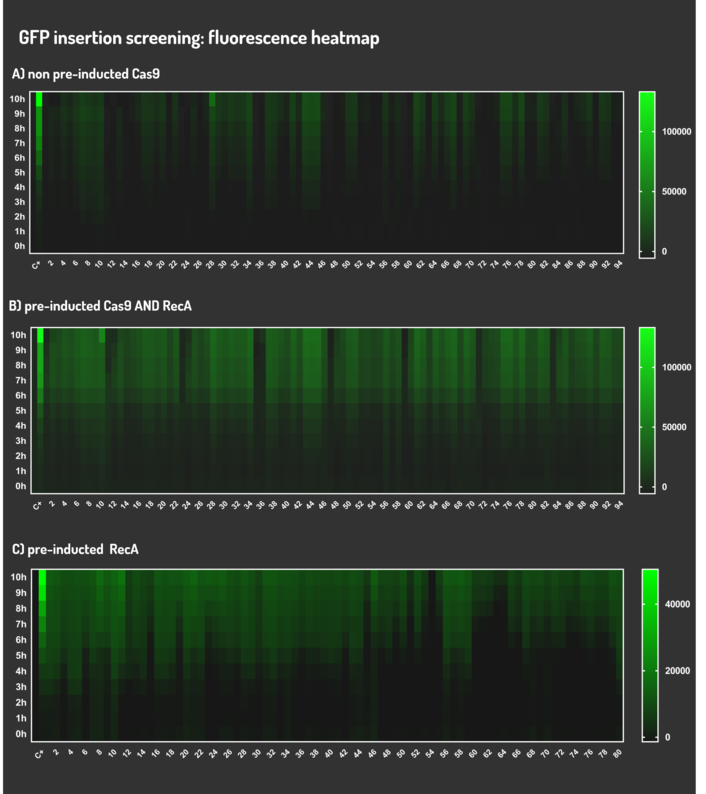
| + | <p>We transformed through electroporation <i>E. coli</i> K12 MG1655 carrying a non-pre-inducted pBAD_Cas9_sgRNA plasmid CamR+ (BBa_K2457006) with pRecA (AmpR+) and a linearized Donor DNA (BBa_K2457004). We screened over 94 transformants colonies, inoculating in 200µL of LB + 25µg/mL chloramphenicol + 100µg/mL ampicillin + 0.2% L-Arabinose + 1mM IPTG to activate the expression of RecA AND Cas9. We measured the initial OD (600nm) and initial fluorescence (exc. 340 nm em. 530 nm) on 96-wells plates applying the Hidex Chameleon V spectrofluorimeter. The process was repeated for each hour over 10 hours while the 96-wells plate kept growing on a shaker at 37ºC.</p> | ||
| + | |||
| + | <p><b>B) carrying a pre-inducted Cas9 + sgRNA AND RecA;</b></p> | ||
| + | |||
| + | <p>We transformed through electroporation <i>E. coli</i> K12 MG1655 carrying a pre-inducted pBAD_Cas9_sgRNA plasmid CamR+ (BBa_K2457006) AND pRecA plasmid AmpR+ with a linearized Donor DNA (BBa_K2457004) + pKana. We screened over 94 transformants colonies, inoculating in 200µL of LB + 50µg/mL kanamycin + 25µg/mL chloramphenicol + 100µg/mL ampicillin + 0.2% L-Arabinose + 1mM IPTG to activate the expression of RecA AND Cas9. We measured the initial OD (600nm) and initial fluorescence (exc. 340 nm em. 530 nm) on 96-wells plates applying the Hidex Chameleon V spectrofluorimeter. The process was repeated for each hour over 10 hours while the 96-wells plate kept growing on a shaker at 37ºC.</p> | ||
| + | |||
| + | <p><b>C) carrying a pre-inducted RecA.</b></p> | ||
| + | <p>We transformed through electroporation <i>E. coli</i> K12 MG1655 carrying a pRecA (AmpR+) with pBAD_Cas9_sgRNA plasmid CamR+ (BBa_K2457006) and a linearized Donor DNA (BBa_K2457004). We screened over 80 transformants colonies, inoculating in 200µL of LB + 25µg/mL chloramphenicol + 100µg/mL ampicillin + 0.2% L-Arabinose + 1mM IPTG to activate the expression of RecA AND Cas9. We measured the initial OD (600nm) and initial fluorescence (exc. 340 nm em. 530 nm) on 96-wells plates applying the Hidex Chameleon V spectrofluorimeter. The process was repeated for each hour over 10 hours while the 96-wells plate kept growing on a shaker at 37ºC.</p> | ||
| + | |||
| + | |||
| + | [[File:CRIN 1.png]] | ||
| + | |||
| + | [[File:CRIN 2.png]] | ||
| + | |||
| + | [[File:CRIN 3.png]] | ||
| + | <p>Figure 7: CRISPeasy-in results.</p> | ||
| + | |||
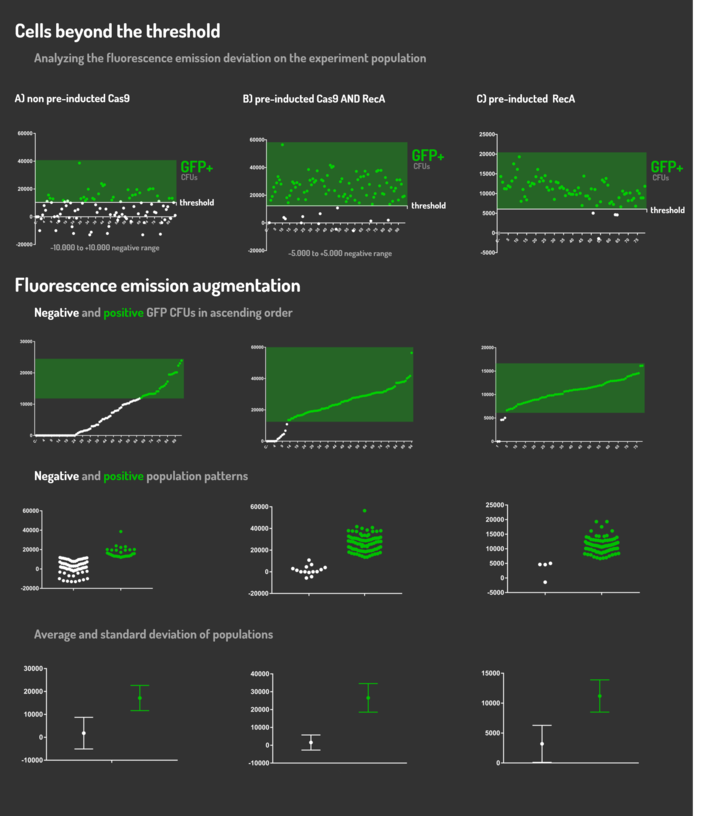
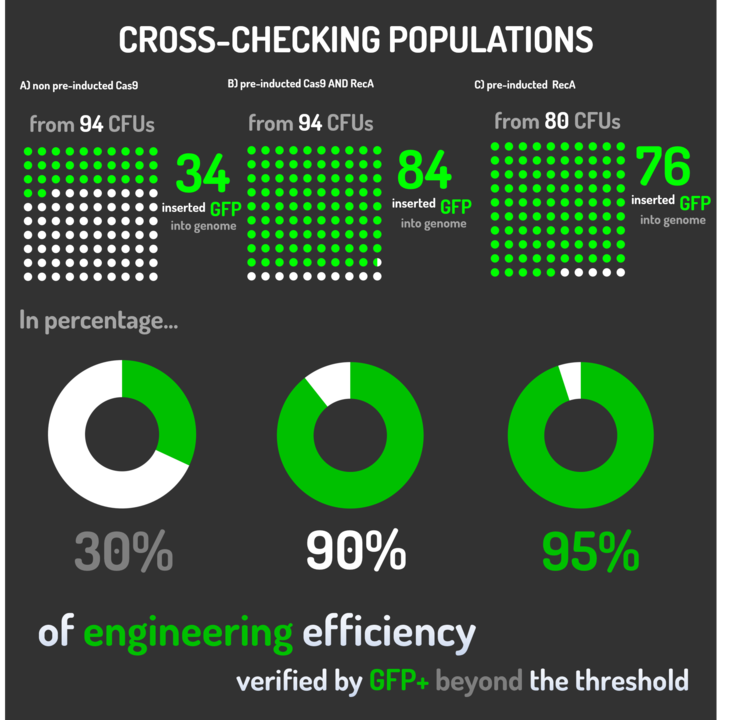
| + | <p>Leading from the achieved results, it was noted that has certain variations between the methodologies performances: with non-induced Cas9 (30% recombinants), with pre-induced Cas9 and RecA (90% recombinants) and with exclusively pre-induced RecA (95% recombinants). </p> | ||
| + | <p>The non-induced Cas9 method possibly do not have the best performance (30%) due to the absence of Cas9 protein when the Donor DNA is inserted into genome, causing a delay in the target sequence cleavage by Cas9, and, therefore, also the delay in homologous recombination, then the major linear Donor DNA is degraded in the bacterial cytosol.</p> | ||
| + | <p>The pre-induced Cas9 and RecA presented a very good result and similar to the most effective approach. When Donor DNA is inserted into the genome, is already happening the cleavage by Cas9 and RecA is investigating a homologous sequence to the broken strand, then Donor DNA is quickly incorporated, having less DNA degradation. </p> | ||
| + | <p>We found out that the most efficient method was the one which had only RecA pre-induced, since Cas9 presence affects the cells growth, therefore, when Cas9 and Donor DNA are added simultaneously, it will have the normal cell growth, beyond the further homologous recombination. </p> | ||
| + | <p>The CRISPeasy framework development can be a boost for an easily AND reliable way to use bacterial genome editing based on CRISPR/Cas9. We encourage teams to dive into CRISPR's standardization with us and expect to be able to see in the next years more and more projects with technique's improvement!</p> | ||
| + | ===Sequencing=== | ||
[[File:BBa K2457002 electropherogram.png]] | [[File:BBa K2457002 electropherogram.png]] | ||
| − | Figure | + | Figure 8: Sequencing electropherogram from BBa_K2457002. |
[[File:BBa K2457002 alignment.png]] | [[File:BBa K2457002 alignment.png]] | ||
| − | Figure | + | Figure 9: Alignment of the designed sequence and our final construction from BBa_K2457002. |
Latest revision as of 03:39, 2 November 2017
Optimized sgRNA targeting LacZ
BBa_K2457002 was engineered to be a building block of the standard BioBrick toolbox for bacterial genome: the ([http://2017.igem.org/Team:Amazonas_Brazil pCRISPeasy]). It’s the first BioBrick available on Registry with an improved sgRNA sequence - both crRNA design and tracrRNA secondary structure - to increase the CRISPR-Cas9 editing efficiency. BBa_K2457002 is composed by: the J23100 constitutive promoter sequence + an optimized sgRNA (crRNA with a GG motif at the 3’ end of it’s target-specific sequence from LacZ [1] and tracrRNA with extended duplex length and a mutation at the thymine 4 [2]) + L3S2P21 terminator [3]. It was rationally designed to be standardized AND interchangeable, aiming to provide a BioBrick to attend multiple genome editing purposes. For that matter, It’s easy to engineer new modules aiming to insert information into the genome through RFC10 Assembly.
Figure 1: BBa_K2457002 circuit.
Figura 2: BBa_K2457002 structure.
Usage and Biology
Single-guide RNA (sgRNA) is one of the two building blocks of the CRISPR-Cas9 genome editing machinery, which directs the Cas9 protein to introduce double-stranded breaks (DSB) into target genomes sequences.
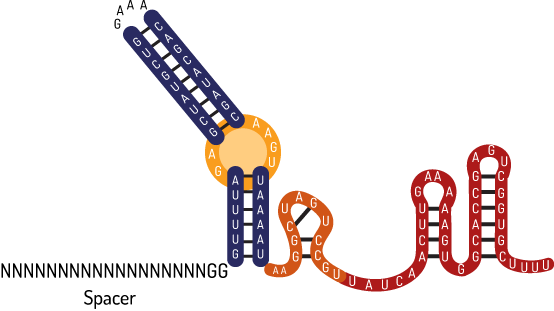
In nature, after the memory development fase made by Cas proteins, the CRISPR locus is transcribed by RNA polymerase synthesizing the crRNA precursor.The post-transcriptional maturation process begins, when this pre-crRNA ,through the complementarity its palindromic repetition, hybridizes itself with a transactivator RNA (tracrRNA), catalyzing the double strand ribonuclease formation RNAses III with the Cas9 protein. This complex cleaves the upstream palindromic repeated hairpin, making small hybridized crRNAs with the transactivator RNA (tracrRNA) in a duo composed by crRNA-tracrRNA, carrying a specific protospacer (complementary sequence) from the invasor DNA. This system, demonstrated on Figure 2 below, Deltcheva et al suggested in 2011, having as inspiration the microRNAs by interference RNAs in eukaryotes.
Therefore, the complex crRNA-tracrRNA consists of: a crRNA, with 42 nucleotides, deriving from the pre-crRNA transcribed on locus CRISPR; and a tracrRNA with 89 nucleotides, transcribed upstream to Cas operon. This non-coding RNAs hybridize themselves by Rosalind-Watson-Crick base pairing, composing a more negatively charged structure. Through polar interaction and bases stacking, the crRNA-tracrRNA is adjusted in a positive charged site between REC and NUC lobuses interface originated at Cas9 protein stemming from Streptococcus pyogenes, changing its quaternary structure to a catalytically active ribonucleoprotein complex (RNP). It has the ability to cleave DNA target sequences that are complementary to the 20 guide nucleotides present in crRNA through the HNH and RuvC nuclease domains.
Design
Synthetic sgRNA structures
Nowadays, the first synthetic chimera of native guide RNA, built by Jinek et al in 2012, is the RNA structure commonly applied in CRISPR Cas9-based scientific projects. This structure has a shorter duplex when compared with native (Fig. 4) composed by: I) a guide sequence with 20 bp + linker loop GAAA + 30 bp of RNA-transactivator tracrRNA.
However, although this structure widely used containing 30 bp of tracrRNA tail is the minimum region for Cas9 interaction, in order to form its ribonucleoprotein complex catalytically active (RNP) in vitro. Guide RNAs, as an extended tail (+67 till +87), drastically enhance Cas9 activity in vivo (Dang et al. 2015)
Assembly
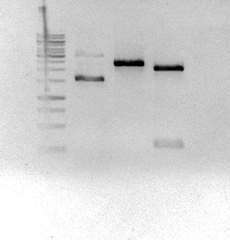
The sequence was ordered as gBlocks from Integrated DNA Technologies and it was assembled by restriction enzyme site.
Figure 3: Eletrophoretic profile of restriction digestion of BBa_K2457002.
Characterization
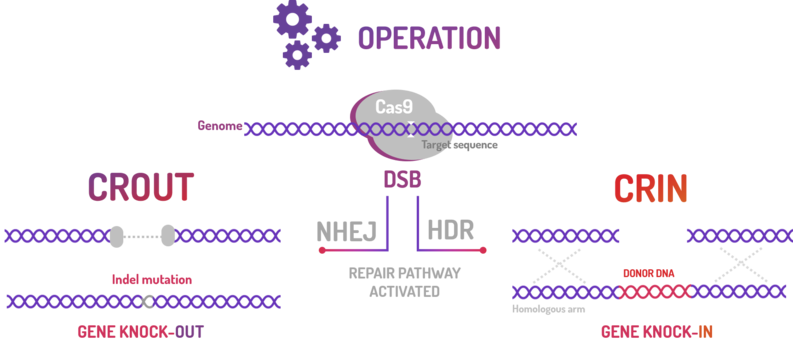
BBa_K2457002 was used by Amazonas_Brazil team in two methods based on standard BioBricks modules for bacterial genome engineering mediated by CRISPR-Cas9 machinery: the CRISPeasys. The approaches constructed were 1) for non-homologous end joining (NHEJ) repair pathway, the CRISPeasy-out method (or crOUT), referring to knock-out and the 2) for homologous directed repair (HDR), the CRISPeasy-in method (or crIN), referring to knock-in.
Figure 4: CRISPeasy subdivisions.
1. CROUT (CRISPeasy-out method): Designed for knock-out strategy for bacterial genome editing based on CRISPR/Cas9
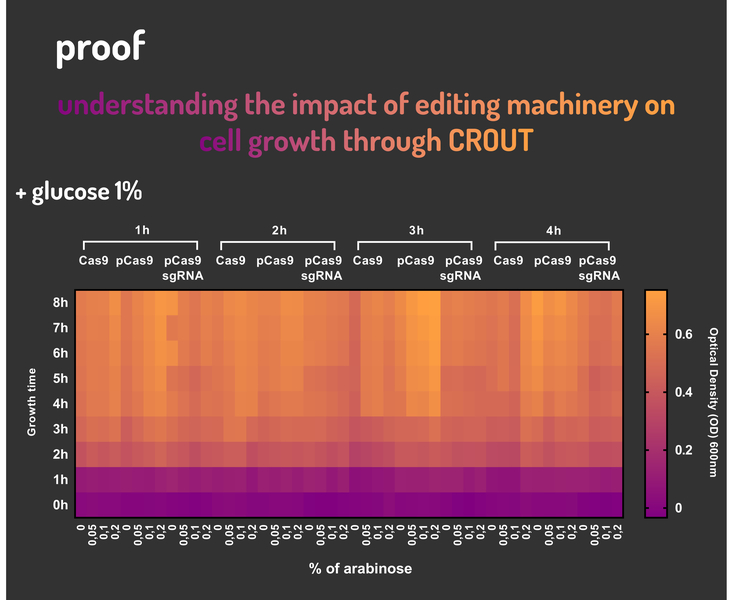
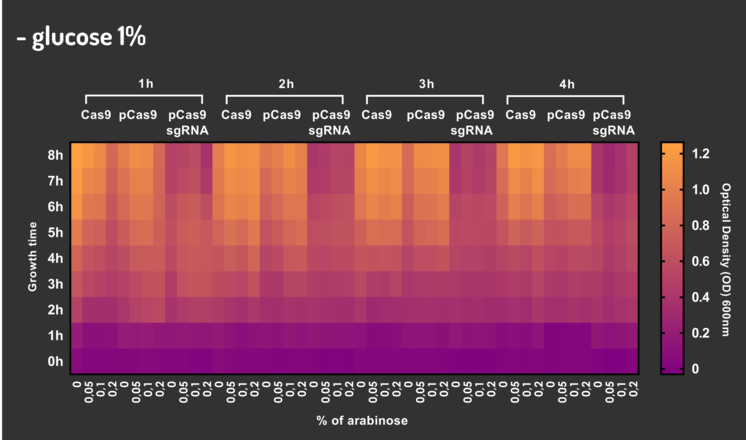
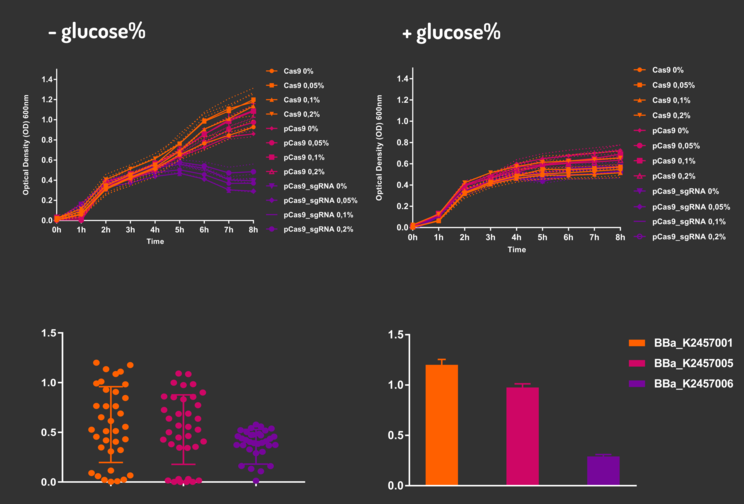
In order to characterize our genetic modules, It was transformed by electroporation E. coli K12 MG1655 with BBa_K2457006 (pBAD_Cas9_sgRNA). We took isolated colonies to pre-inoculate in 5mL of Luria-Bertani (LB), incubating overnight in a shaker at 37ºC. After that, we measured the Optical Density (OD) on spectrophotometer 600 nm aiming to standardize the cell ratio to 1 OD. All the samples were inoculated to 0.05 OD. Our experiment was performed in 96-well plates with 200µL of LB + 25µg/mL chloramphenicol, measuring the cell growth for over 8h in a range of conditions (see the layout plate below). We implemented Hidex Chameleon spectrophotometer to measure the OD and incubate the 96-well plates into shaker at 37°C. As a control, we employed MG1655 carrying two genetic circuits: BBa_K2457001 (Standardized Cas9 coding sequence without regulatory modules) AND BBa_K2457005 (Cas9 device without sgRNA).
Glucose 1% was applied as a repressor to understand the activation and deactivation of the Cas9 expression and was added in 50% of the samples. The L-Arabinose solution is used as the inductor of the AraC_pBAD regulatory module controlling the Cas9 expression and was added in 75% of the samples, on respective concentrations: 0,05%, 0,1% and 0,2%. It was added at 1, 2, 3 and 4 hours of cell growth.
Figure 5: CRISPeasy-out results.
Based on a range of experiments involving the knocking-out process, we could propose that in the glucose (repressor) absence AND in the arabinose presence (inductor), MG1655 transformed with the BBa_K2457006 vector, carrying the Cas9 device and constitutively expressing sgRNA, reported a meaningful lower growth in contrast with the samples without the regulatory module (BBa_K2457001) OR without the sgRNA (BBa_K2457005). And, in the glucose presence, repressing the Cas9 expression, all the samples displayed a similar growth performance. The samples which carried the sgRNA (i.e., directing the Cas9 sequence to lead the DSB into a specific point of the genome) presented a low cell viability, supporting the hypothesis that even occurring the Non-Homologous End Joining (NHEJ) cell repair, not all cells could recover themselves, leading to the cellular death. Thus, we founded our proof of concept on the well-characterized impact caused by the Cas9 activity into the cellular machinery AND cell growth.
2. CRIN (CRISPeasy-in method): Designed for knock-in strategy for bacterial genome editing based on CRISPR/Cas9
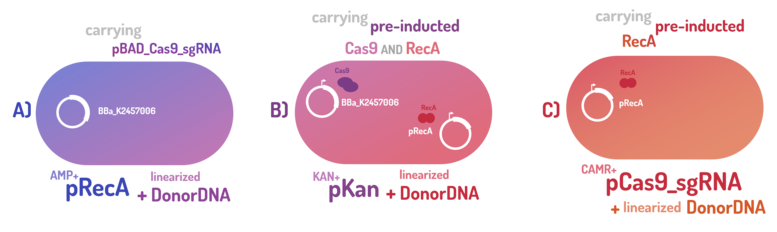
As the main key to this method, there is an AND logic gate composed of Cas9 coupled with RecA, which operates the Donor DNA insertion into E. coli genome through Direct Homology Repair (HDR). In this way, the arabinose AND IPTG (the inputs) activate the gene expression program encoding Cas9 and RecA. Then, there is the execution of their hardware's (physical parts, e.g. proteins) operation. As an output, Cas9 AND RecA activate the DNA insertion into the genome. In order to standardize a reliable datasheet of genome engineering efficiency, we tested a range of electrocompetent E. coli K12 MG1655 cells with different circuits: test A) carrying a non-pre-inducted Cas9 device + sgRNA (BBa_K2457006) plasmid; B) carrying a pre-inducted Cas9 AND RecA; C) carrying a pre-inducted RecA. The donor DNA was electroporated in all the tests.
Figure 6: CRISPeasy-in methodology.
A) carrying a non-pre-inducted Cas9 device + sgRNA;
We transformed through electroporation E. coli K12 MG1655 carrying a non-pre-inducted pBAD_Cas9_sgRNA plasmid CamR+ (BBa_K2457006) with pRecA (AmpR+) and a linearized Donor DNA (BBa_K2457004). We screened over 94 transformants colonies, inoculating in 200µL of LB + 25µg/mL chloramphenicol + 100µg/mL ampicillin + 0.2% L-Arabinose + 1mM IPTG to activate the expression of RecA AND Cas9. We measured the initial OD (600nm) and initial fluorescence (exc. 340 nm em. 530 nm) on 96-wells plates applying the Hidex Chameleon V spectrofluorimeter. The process was repeated for each hour over 10 hours while the 96-wells plate kept growing on a shaker at 37ºC.
B) carrying a pre-inducted Cas9 + sgRNA AND RecA;
We transformed through electroporation E. coli K12 MG1655 carrying a pre-inducted pBAD_Cas9_sgRNA plasmid CamR+ (BBa_K2457006) AND pRecA plasmid AmpR+ with a linearized Donor DNA (BBa_K2457004) + pKana. We screened over 94 transformants colonies, inoculating in 200µL of LB + 50µg/mL kanamycin + 25µg/mL chloramphenicol + 100µg/mL ampicillin + 0.2% L-Arabinose + 1mM IPTG to activate the expression of RecA AND Cas9. We measured the initial OD (600nm) and initial fluorescence (exc. 340 nm em. 530 nm) on 96-wells plates applying the Hidex Chameleon V spectrofluorimeter. The process was repeated for each hour over 10 hours while the 96-wells plate kept growing on a shaker at 37ºC.
C) carrying a pre-inducted RecA.
We transformed through electroporation E. coli K12 MG1655 carrying a pRecA (AmpR+) with pBAD_Cas9_sgRNA plasmid CamR+ (BBa_K2457006) and a linearized Donor DNA (BBa_K2457004). We screened over 80 transformants colonies, inoculating in 200µL of LB + 25µg/mL chloramphenicol + 100µg/mL ampicillin + 0.2% L-Arabinose + 1mM IPTG to activate the expression of RecA AND Cas9. We measured the initial OD (600nm) and initial fluorescence (exc. 340 nm em. 530 nm) on 96-wells plates applying the Hidex Chameleon V spectrofluorimeter. The process was repeated for each hour over 10 hours while the 96-wells plate kept growing on a shaker at 37ºC.
Figure 7: CRISPeasy-in results.
Leading from the achieved results, it was noted that has certain variations between the methodologies performances: with non-induced Cas9 (30% recombinants), with pre-induced Cas9 and RecA (90% recombinants) and with exclusively pre-induced RecA (95% recombinants).
The non-induced Cas9 method possibly do not have the best performance (30%) due to the absence of Cas9 protein when the Donor DNA is inserted into genome, causing a delay in the target sequence cleavage by Cas9, and, therefore, also the delay in homologous recombination, then the major linear Donor DNA is degraded in the bacterial cytosol.
The pre-induced Cas9 and RecA presented a very good result and similar to the most effective approach. When Donor DNA is inserted into the genome, is already happening the cleavage by Cas9 and RecA is investigating a homologous sequence to the broken strand, then Donor DNA is quickly incorporated, having less DNA degradation.
We found out that the most efficient method was the one which had only RecA pre-induced, since Cas9 presence affects the cells growth, therefore, when Cas9 and Donor DNA are added simultaneously, it will have the normal cell growth, beyond the further homologous recombination.
The CRISPeasy framework development can be a boost for an easily AND reliable way to use bacterial genome editing based on CRISPR/Cas9. We encourage teams to dive into CRISPR's standardization with us and expect to be able to see in the next years more and more projects with technique's improvement!
Sequencing
 Figure 8: Sequencing electropherogram from BBa_K2457002.
Figure 8: Sequencing electropherogram from BBa_K2457002.
Figure 9: Alignment of the designed sequence and our final construction from BBa_K2457002.
Sequence and Features
- 10COMPATIBLE WITH RFC[10]
- 12COMPATIBLE WITH RFC[12]
- 21COMPATIBLE WITH RFC[21]
- 23COMPATIBLE WITH RFC[23]
- 25COMPATIBLE WITH RFC[25]
- 1000COMPATIBLE WITH RFC[1000]