Difference between revisions of "Part:BBa K404113"
(→Additional characterization of BBa_K404113 by Munich 2016) |
|||
| (23 intermediate revisions by 3 users not shown) | |||
| Line 1: | Line 1: | ||
__NOTOC__ | __NOTOC__ | ||
<partinfo>BBa_K404113 short</partinfo> | <partinfo>BBa_K404113 short</partinfo> | ||
| + | |||
| + | =Additional characterization of BBa_K404113 by [http://2016.igem.org/Team:LMU-TUM_Munich Munich 2016]= | ||
| + | |||
| + | [[File:Muc16_Killswicht_001.png |thumb|center|900px| ]] | ||
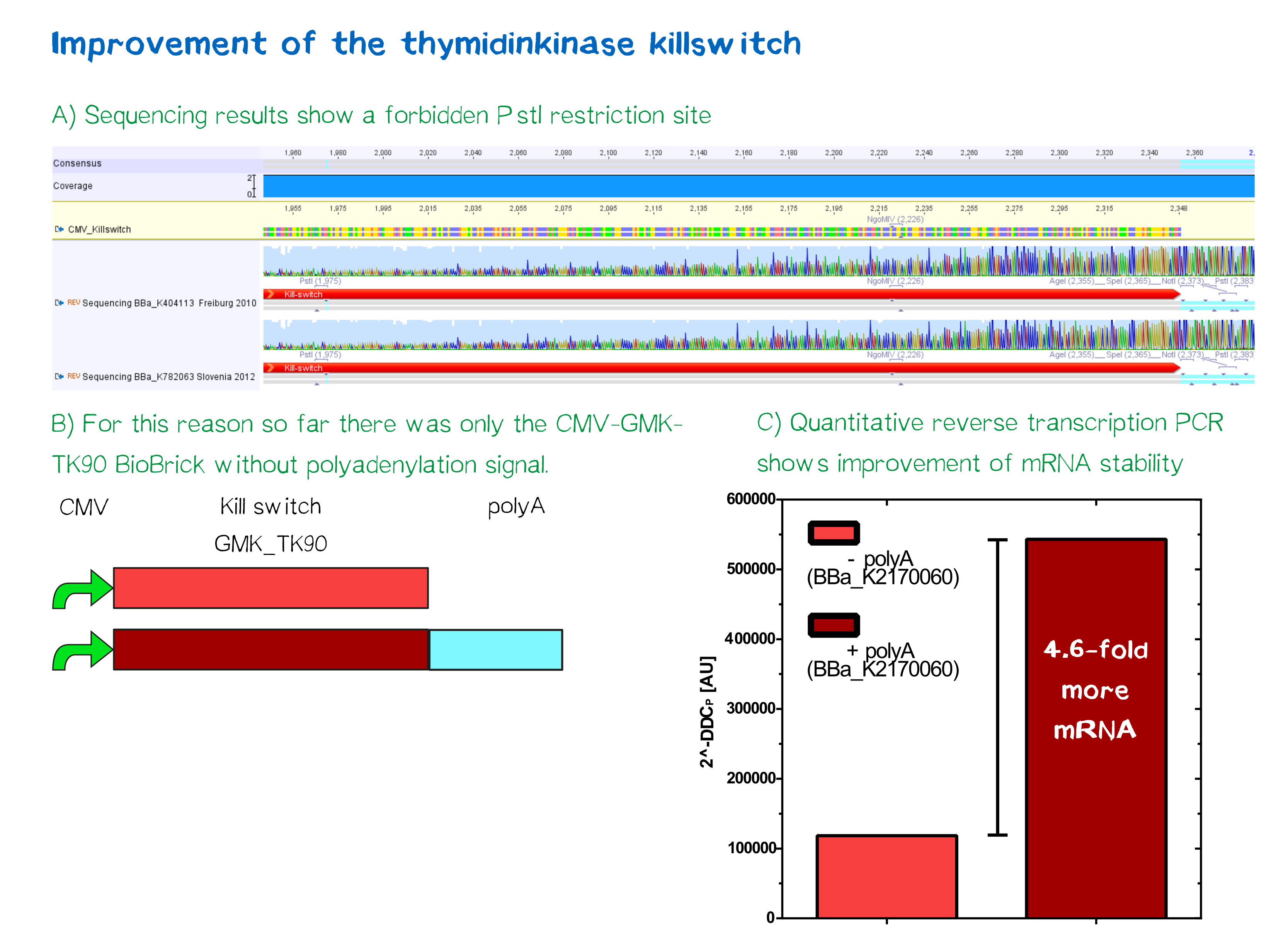
| + | This part '''contains a forbidden PstI site''', which is generated by a point mutation in position 1.309. This restriction site makes a ligation of a BioBrick in the donstream region of this Biobrick impossible. This is why we created the RFC[10] compatible BioBrick [https://parts.igem.org/wiki/index.php?title=Part:BBa_K2170150 BBa_K2170150] (pCMV-GMK_TK90), which carries a CMV promotor in the upstream region of this BioBrick and [https://parts.igem.org/wiki/index.php?title=Part:BBa_K2170060 BBa_K2170060] (pCMV-GMK_TK90-polyA), that contains an additional poly adenylation site for improved mRNA stability. These improved parts are characterized with respect to mRNA expression level and functionality in cell culture. | ||
| + | |||
| + | Quantitative analysis by mRNA measuring of Poly-A-sequence addition showed 4.6-fold higher mRNA stability in comparison to the non-Poly-A-construct. [http://2016.igem.org/Team:LMU-TUM_Munich/Safety More information]<br> | ||
| + | <hr> | ||
| + | |||
| + | |||
[[Image:Freiburg10_VectorplasmidBricks 10.png|thumb|center|480px]]<br> | [[Image:Freiburg10_VectorplasmidBricks 10.png|thumb|center|480px]]<br> | ||
| − | <h3>Tymidine kinase</h3> | + | |
| + | <h3>Tymidine kinase </h3>(BBa_K404109)<br> | ||
'''Thymidine kinase (TK)''' (EC 2.7.1.21) is known to be involved in the salvage pathway of nucleosides to nucleotides (Andrei et al. 2005). | '''Thymidine kinase (TK)''' (EC 2.7.1.21) is known to be involved in the salvage pathway of nucleosides to nucleotides (Andrei et al. 2005). | ||
Due to its broader spectrum for different substrates, herpes simplex virus thymidine kinase (HSV-TK) is widely used in gene therapy approaches instead of endogenous thymidine kinases (Black et al. 1996). The transgenic introduced HSV-TK monophosphrylates nucleosides or nucleoside analogs such as ganciclovir (GCV) or acyclovir (AVC) followed by further phosphorylation through cellular kinases to nucleoside triphsphosphates. Incorporation of nucleotide analogs such as ganciclovir triphosphate or acyclovir triphosphates leads to DNA chain termination (Reardon 1989) and finally results in cell death. | Due to its broader spectrum for different substrates, herpes simplex virus thymidine kinase (HSV-TK) is widely used in gene therapy approaches instead of endogenous thymidine kinases (Black et al. 1996). The transgenic introduced HSV-TK monophosphrylates nucleosides or nucleoside analogs such as ganciclovir (GCV) or acyclovir (AVC) followed by further phosphorylation through cellular kinases to nucleoside triphsphosphates. Incorporation of nucleotide analogs such as ganciclovir triphosphate or acyclovir triphosphates leads to DNA chain termination (Reardon 1989) and finally results in cell death. | ||
Genetic modifications of the active site represented by a tripeptide motif in thymidine kinase increases the substrate affinity of HSV-TK towards GCV and ACV (Black et al. 1996). Two promising mutant HSV-TKs have been found by large mutagenesis screenings modifying several amino acids and conducting sensitivity assays for ganciclovir and acyclovir (Black et al. 2001). | Genetic modifications of the active site represented by a tripeptide motif in thymidine kinase increases the substrate affinity of HSV-TK towards GCV and ACV (Black et al. 1996). Two promising mutant HSV-TKs have been found by large mutagenesis screenings modifying several amino acids and conducting sensitivity assays for ganciclovir and acyclovir (Black et al. 2001). | ||
<br> | <br> | ||
| − | <p><h3>Mouse guanlyate kinase</h3> | + | <p><h3>Mouse guanlyate kinase </h3>(BBa_K404111)<br> |
<b>Guanylate kinases (GMKs)</b> are involved in the salvage pathway of mono-phosphorylated guanosine (GMP) nucleosides to GDP therefore being essential in nucleotide maturation (Stolworthy & Black 2001). By introducing transgenic thymidine kinases (TKs) into tumor cells, a bottleneck occurs by overexpression of mono-phosphorylated intermediates. To overcome the accumulation of these non-toxic molecules, Willmon, Krabbenhoft, & Black (2006) fused the herpes simplex virus thymidine kinase (HSV-TK) to the guanylate kinase from M. musculus and demonstrated enhanced tumor killing in vitro. <br /> | <b>Guanylate kinases (GMKs)</b> are involved in the salvage pathway of mono-phosphorylated guanosine (GMP) nucleosides to GDP therefore being essential in nucleotide maturation (Stolworthy & Black 2001). By introducing transgenic thymidine kinases (TKs) into tumor cells, a bottleneck occurs by overexpression of mono-phosphorylated intermediates. To overcome the accumulation of these non-toxic molecules, Willmon, Krabbenhoft, & Black (2006) fused the herpes simplex virus thymidine kinase (HSV-TK) to the guanylate kinase from M. musculus and demonstrated enhanced tumor killing in vitro. <br /> | ||
<br /> | <br /> | ||
| − | + | ||
<!-- Add more about the biology of this part here | <!-- Add more about the biology of this part here | ||
| Line 25: | Line 36: | ||
===Functional Parameters=== | ===Functional Parameters=== | ||
<partinfo>BBa_K404113 parameters</partinfo> | <partinfo>BBa_K404113 parameters</partinfo> | ||
| − | <!-- --> | + | <!-- --> <br> |
| + | |||
| + | [http://2012.igem.org/Team:Slovenia Team Slovenia 2012] | ||
| + | further characterized and improved fusion protein mGMK_TK30 by adding CMV promoter. Check results on [https://parts.igem.org/Part:BBa_K404113:Experience Experience]. | ||
| + | |||
| + | |||
| + | |||
| + | <h3>References</h3> | ||
| + | <b>Andrei G, Balzarini J, Fiten P</b> "Characterization of herpes simplex virus type 1 thymidine kinase mutants selected under a single round of high-dose brivudin." Journal of virology. 2005;79(9):5863-9.<br /> | ||
| + | <b> Ardiani a, Sanchez-Bonilla M, Black ME</b> " Fusion enzymes containing HSV-1 thymidine kinase mutants and guanylate kinase enhance prodrug sensitivity in vitro and in vivo." Cancer gene therapy. 2010;17(2):86-96<br /> | ||
| + | <b>Black ME, Newcomb TG, Wilson HM, Loeb La.</b> "Creation of drug-specific herpes simplex virus type 1 thymidine kinase mutants for gene therapy." Proceedings of the National Academy of Sciences of the United States of America. 1996;93(8):3525-9<br /> | ||
| + | <b>Black ME, Kokoris MS, Sabo P.</b> "Herpes simplex virus-1 thymidine kinase mutants created by semi-random sequence mutagenesis improve prodrug-mediated tumor cell killing." Cancer research. 2001;61(7):3022-6 <br /> | ||
| + | <b>Sekulic, N. et al.</b>, 2002. Structural characterization of the closed conformation of mouse guanylate kinase. The Journal of biological chemistry, 277(33), 30236-43. Available at: http://www.ncbi.nlm.nih.gov/pubmed/12036965. | ||
| + | <br /> | ||
| + | <b>Stolworthy, T.S. & Black, M.E.</b>, 2001. The mouse guanylate kinase double mutant E72Q/D103N is a functional adenylate kinase. Protein engineering, 14(11), 903-9. Available at: http://www.ncbi.nlm.nih.gov/pubmed/11742110. | ||
| + | <br /> | ||
| + | <b>Willmon, C.L., Krabbenhoft, E. & Black, M.E.</b>, 2006. A guanylate kinase/HSV-1 thymidine kinase fusion protein enhances prodrug-mediated cell killing. Gene therapy, 13(17), 1309-12. Available at: http://www.ncbi.nlm.nih.gov/pubmed/16810197. | ||
Latest revision as of 23:30, 19 October 2016
Mouse guanylate kinase - thymidine kinase TK30 (Fusion protein mGMK_TK30)
Additional characterization of BBa_K404113 by [http://2016.igem.org/Team:LMU-TUM_Munich Munich 2016]
This part contains a forbidden PstI site, which is generated by a point mutation in position 1.309. This restriction site makes a ligation of a BioBrick in the donstream region of this Biobrick impossible. This is why we created the RFC[10] compatible BioBrick BBa_K2170150 (pCMV-GMK_TK90), which carries a CMV promotor in the upstream region of this BioBrick and BBa_K2170060 (pCMV-GMK_TK90-polyA), that contains an additional poly adenylation site for improved mRNA stability. These improved parts are characterized with respect to mRNA expression level and functionality in cell culture.
Quantitative analysis by mRNA measuring of Poly-A-sequence addition showed 4.6-fold higher mRNA stability in comparison to the non-Poly-A-construct. [http://2016.igem.org/Team:LMU-TUM_Munich/Safety More information]
Tymidine kinase
(BBa_K404109)Thymidine kinase (TK) (EC 2.7.1.21) is known to be involved in the salvage pathway of nucleosides to nucleotides (Andrei et al. 2005).
Due to its broader spectrum for different substrates, herpes simplex virus thymidine kinase (HSV-TK) is widely used in gene therapy approaches instead of endogenous thymidine kinases (Black et al. 1996). The transgenic introduced HSV-TK monophosphrylates nucleosides or nucleoside analogs such as ganciclovir (GCV) or acyclovir (AVC) followed by further phosphorylation through cellular kinases to nucleoside triphsphosphates. Incorporation of nucleotide analogs such as ganciclovir triphosphate or acyclovir triphosphates leads to DNA chain termination (Reardon 1989) and finally results in cell death.
Genetic modifications of the active site represented by a tripeptide motif in thymidine kinase increases the substrate affinity of HSV-TK towards GCV and ACV (Black et al. 1996). Two promising mutant HSV-TKs have been found by large mutagenesis screenings modifying several amino acids and conducting sensitivity assays for ganciclovir and acyclovir (Black et al. 2001).
Mouse guanlyate kinase
(BBa_K404111)Guanylate kinases (GMKs) are involved in the salvage pathway of mono-phosphorylated guanosine (GMP) nucleosides to GDP therefore being essential in nucleotide maturation (Stolworthy & Black 2001). By introducing transgenic thymidine kinases (TKs) into tumor cells, a bottleneck occurs by overexpression of mono-phosphorylated intermediates. To overcome the accumulation of these non-toxic molecules, Willmon, Krabbenhoft, & Black (2006) fused the herpes simplex virus thymidine kinase (HSV-TK) to the guanylate kinase from M. musculus and demonstrated enhanced tumor killing in vitro.
Sequence and Features
- 10COMPATIBLE WITH RFC[10]
- 12COMPATIBLE WITH RFC[12]
- 21COMPATIBLE WITH RFC[21]
- 23COMPATIBLE WITH RFC[23]
- 25INCOMPATIBLE WITH RFC[25]Illegal NgoMIV site found at 653
Illegal NgoMIV site found at 1558 - 1000INCOMPATIBLE WITH RFC[1000]Illegal SapI.rc site found at 45
[http://2012.igem.org/Team:Slovenia Team Slovenia 2012] further characterized and improved fusion protein mGMK_TK30 by adding CMV promoter. Check results on Experience.
References
Andrei G, Balzarini J, Fiten P "Characterization of herpes simplex virus type 1 thymidine kinase mutants selected under a single round of high-dose brivudin." Journal of virology. 2005;79(9):5863-9.
Ardiani a, Sanchez-Bonilla M, Black ME " Fusion enzymes containing HSV-1 thymidine kinase mutants and guanylate kinase enhance prodrug sensitivity in vitro and in vivo." Cancer gene therapy. 2010;17(2):86-96
Black ME, Newcomb TG, Wilson HM, Loeb La. "Creation of drug-specific herpes simplex virus type 1 thymidine kinase mutants for gene therapy." Proceedings of the National Academy of Sciences of the United States of America. 1996;93(8):3525-9
Black ME, Kokoris MS, Sabo P. "Herpes simplex virus-1 thymidine kinase mutants created by semi-random sequence mutagenesis improve prodrug-mediated tumor cell killing." Cancer research. 2001;61(7):3022-6
Sekulic, N. et al., 2002. Structural characterization of the closed conformation of mouse guanylate kinase. The Journal of biological chemistry, 277(33), 30236-43. Available at: http://www.ncbi.nlm.nih.gov/pubmed/12036965.
Stolworthy, T.S. & Black, M.E., 2001. The mouse guanylate kinase double mutant E72Q/D103N is a functional adenylate kinase. Protein engineering, 14(11), 903-9. Available at: http://www.ncbi.nlm.nih.gov/pubmed/11742110.